Lipid-dependent Akt-ivity: where, when, and how
- PMID: 31147387
- PMCID: PMC6599160
- DOI: 10.1042/BST20190013
Lipid-dependent Akt-ivity: where, when, and how
Abstract
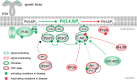
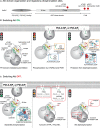
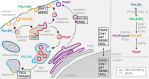
Akt is an essential protein kinase activated downstream of phosphoinositide 3-kinase and frequently hyperactivated in cancer. Canonically, Akt is activated by phosphoinositide-dependent kinase 1 and mechanistic target of rapamycin complex 2, which phosphorylate it on two regulatory residues in its kinase domain upon targeting of Akt to the plasma membrane by PI(3,4,5)P3 Recent evidence, however, has shown that, in addition to phosphorylation, Akt activity is allosterically coupled to the engagement of PI(3,4,5)P3 or PI(3,4)P2 in cellular membranes. Furthermore, the active membrane-bound conformation of Akt is protected from dephosphorylation, and Akt inactivation by phosphatases is rate-limited by its dissociation. Thus, Akt activity is restricted to membranes containing either PI(3,4,5)P3 or PI(3,4)P2 While PI(3,4,5)P3 has long been associated with signaling at the plasma membrane, PI(3,4)P2 is gaining increasing traction as a signaling lipid and has been implicated in controlling Akt activity throughout the endomembrane system. This has clear implications for the phosphorylation of both freely diffusible substrates and those localized to discrete subcellular compartments.
Keywords: 4; 4)P2; 5)P3; Akt; PI(3; kinase; lipids; phosphorylation/dephosphorylation.
© 2019 The Author(s).
Conflict of interest statement
The Authors declare that there are no competing interests associated with the manuscript.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

