Classical and novel strategies to develop a Shigella glycoconjugate vaccine: from concept to efficacy in human
- PMID: 31158047
- PMCID: PMC6663142
- DOI: 10.1080/21645515.2019.1606972
Classical and novel strategies to develop a Shigella glycoconjugate vaccine: from concept to efficacy in human
Abstract
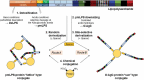
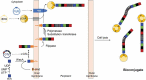
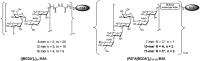
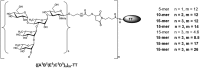
Shigella are gram-negative bacteria that cause severe diarrhea and dysentery, with a high level of antimicrobial resistance. Disease-induced protection against reinfection in Shigella-endemic areas provides convincing evidence on the feasibility of a vaccine and on the importance of Shigella lipopolysaccharides as targets of the host humoral protective immune response against disease. This article provides an overview of the original and current strategies toward the development of a Shigella glycan-protein conjugate vaccine that would cover the most commonly detected strains. Going beyond pioneering "lattice"-type polysaccharide-protein conjugates, progress, and challenges are addressed with focus on promising alternatives, which have reached phases I and II clinical trial. Glycoengineered bioconjugates and "sun"-type conjugates featuring well-defined synthetic carbohydrate antigens are discussed with insights on the molecular parameters governing the rational design of a cost-effective glycoconjugate vaccine efficacious in preventing diseases caused by Shigella in the most at risk populations, young children living in endemic areas.
Keywords: Bioconjugate; O-acetylation; Shigella; carbohydrate antigen; clinical trial; glycan; glycoconjugate; lipopolysaccharide; oligosaccharide; vaccine.
Figures


Similar articles
-
Characterization of functional oligosaccharide mimics of the Shigella flexneri serotype 2a O-antigen: implications for the development of a chemically defined glycoconjugate vaccine.J Immunol. 2006 Feb 1;176(3):1686-94. doi: 10.4049/jimmunol.176.3.1686. J Immunol. 2006. PMID: 16424198
-
Status of vaccine research and development for Shigella.Vaccine. 2016 Jun 3;34(26):2887-2894. doi: 10.1016/j.vaccine.2016.02.075. Epub 2016 Mar 12. Vaccine. 2016. PMID: 26979135 Review.
-
In vivo production of a novel glycoconjugate vaccine against Shigella flexneri 2a in recombinant Escherichia coli: identification of stimulating factors for in vivo glycosylation.Microb Cell Fact. 2015 Jan 23;14:12. doi: 10.1186/s12934-015-0195-7. Microb Cell Fact. 2015. PMID: 25612741 Free PMC article.
-
A Novel Shigella Proteome Microarray Discriminates Targets of Human Antibody Reactivity following Oral Vaccination and Experimental Challenge.mSphere. 2018 Aug 1;3(4):e00260-18. doi: 10.1128/mSphere.00260-18. mSphere. 2018. PMID: 30068560 Free PMC article.
-
Shigella vaccine development: prospective animal models and current status.Curr Pharm Biotechnol. 2013;14(10):903-12. doi: 10.2174/1389201014666131226123900. Curr Pharm Biotechnol. 2013. PMID: 24372251 Review.
Cited by
-
Computational Design of a Chimeric Vaccine against Plesiomonas shigelloides Using Pan-Genome and Reverse Vaccinology.Vaccines (Basel). 2022 Nov 8;10(11):1886. doi: 10.3390/vaccines10111886. Vaccines (Basel). 2022. PMID: 36366394 Free PMC article.
-
Vaccines against gastroenteritis, current progress and challenges.Gut Microbes. 2020 Nov 1;11(6):1486-1517. doi: 10.1080/19490976.2020.1770666. Epub 2020 Jun 18. Gut Microbes. 2020. PMID: 32552414 Free PMC article. Review.
-
Molecular Modeling of the Shigella flexneri Serogroup 3 and 5 O-Antigens and Conformational Relationships for a Vaccine Containing Serotypes 2a and 3a.Vaccines (Basel). 2020 Nov 2;8(4):643. doi: 10.3390/vaccines8040643. Vaccines (Basel). 2020. PMID: 33147882 Free PMC article.
-
Recent advances on smart glycoconjugate vaccines in infections and cancer.FEBS J. 2022 Jul;289(14):4251-4303. doi: 10.1111/febs.15909. Epub 2021 Jun 1. FEBS J. 2022. PMID: 33934527 Free PMC article. Review.
-
The First-in-Human Synthetic Glycan-Based Conjugate Vaccine Candidate against Shigella.ACS Cent Sci. 2022 Apr 27;8(4):449-460. doi: 10.1021/acscentsci.1c01479. Epub 2022 Mar 17. ACS Cent Sci. 2022. PMID: 35559427 Free PMC article.
References
-
- Platts-Mills JA, Babji S, Bodhidatta L, Gratz J, Haque R, Havt A, McCormick BJ, McGrath M, Olortequi MP, Samie A, et al. Pathogenspecific burdens of community diarrhoea in developing countries: a multisite birth cohort study (MAL-ED). Lancet Glob Health. 2015;3:e564–75. doi:10.1016/S2214-109X(15)00151-5. - DOI - PMC - PubMed
-
- Liu J, Platts-Mills JA, Juma J, Kabir F, Nkeze J, Okoi C, Operario DJ, Uddin J, Ahmed S, Alonso PL, et al. Use of quantitative molecular diagnostic methods to identify causes of diarrhoea in children: a reanalysis of the GEMS case-control study. Lancet. 2016;388:1291–301. doi:10.1016/S0140-6736(16)31529-X. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
