Monocyte Polarization is Altered by Total-Body Irradiation in Male Rhesus Macaques: Implications for Delayed Effects of Acute Radiation Exposure
- PMID: 31161966
- PMCID: PMC6698159
- DOI: 10.1667/RR15310.1
Monocyte Polarization is Altered by Total-Body Irradiation in Male Rhesus Macaques: Implications for Delayed Effects of Acute Radiation Exposure
Abstract
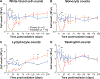
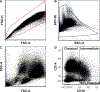
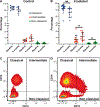
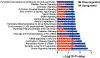
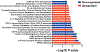
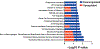
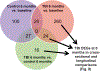
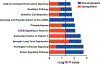
Radiation-induced fibrosis (RIF) is a common delayed effect of acute ionizing radiation exposure (DEARE) affecting diverse tissues including the heart, lungs, liver and skin, leading to reduced tissue function and increased morbidity. Monocytes, which may be classified into classical (CD14++, CD16-), intermediate (CD14++, CD16+) and non-classical (CD14+/low, CD16++) subtypes in humans and non-human primates (NHPs), and monocyte-derived macrophages may play an integral role in the pathogenesis of RIF. We tested the hypothesis that moderate to high levels of total-body exposure to radiation would alter monocyte polarization and produce phenotypes that could promote multi-organ fibrosis in a wellestablished NHP model of DEARE. Subjects were 16 young adult male rhesus macaques, ten of which were exposed to high-energy, 4 Gy X-ray total-body irradiation (TBI) and six that received sham irradiation (control). Total monocytes assessed by complete blood counts were 89% depleted in TBI animals by day 9 postirradiation (P < 0.05), but recovered by day 30 postirradiation and did not differ from control levels thereafter. Monocytes were isolated from peripheral blood mononuclear cells (PBMCs) and sorted into classical, intermediate and non-classical subsets using fluorescence-activated cell sorting (FACS) prior to and at 6 months post-TBI. At 6 months postirradiation, monocyte polarization shifted towards lower classical (92% → 86%) and higher intermediate (7% → 12%) and non-classical monocyte subsets (0.6% → 2%) (all P < 0.05) in TBI animals compared to baseline. No change in monocyte subsets was observed in control animals. Transcriptional profiles in classical and intermediate monocyte subsets were assessed using RNAseq. Classical monocyte gene expression did not change significantly over time or differ cross-sectionally between TBI and control groups. In contrast, significant numbers of differentially expressed genes (DEGs) were detected in intermediate monocyte comparisons between the TBI animals and all animals at baseline (304 DEGs), and in the TBI versus control animals at 6 months postirradiation (67 DEGs). Intermediate monocytes also differed between baseline and 6 months in control animals (147 DEGs). Pathway analysis was used to identify genes within significant canonical pathways, yielding 52 DEGs that were specific to irradiated intermediate monocytes. These DEGs and significant canonical pathways were associated with pro-fibrotic and anti-inflammatory signaling pathways that have been noted to induce M2 macrophage polarization. These findings support the hypothesis that TBI may alter monocyte programming and polarization towards a profibrotic phenotype, providing a novel target opportunity for therapies to inhibit or prevent RIF.
Figures
Similar articles
-
Elevated circulating CD14lowCD16+ monocyte subset in primary biliary cirrhosis correlates with liver injury and promotes Th1 polarization.Clin Exp Med. 2016 Nov;16(4):511-521. doi: 10.1007/s10238-015-0381-2. Epub 2015 Sep 24. Clin Exp Med. 2016. PMID: 26403460
-
Monocyte Subsets Have Distinct Patterns of Tetraspanin Expression and Different Capacities to Form Multinucleate Giant Cells.Front Immunol. 2018 Jun 8;9:1247. doi: 10.3389/fimmu.2018.01247. eCollection 2018. Front Immunol. 2018. PMID: 29937768 Free PMC article.
-
Monocyte subsets exhibit transcriptional plasticity and a shared response to interferon in SIV-infected rhesus macaques.J Leukoc Biol. 2018 Jan;103(1):141-155. doi: 10.1002/JLB.4A0217-047R. Epub 2017 Dec 14. J Leukoc Biol. 2018. PMID: 29345061 Free PMC article.
-
The continuum of monocyte phenotypes: Experimental evidence and prognostic utility in assessing cardiovascular risk.J Leukoc Biol. 2018 Mar 30. doi: 10.1002/JLB.5RU1217-477RR. Online ahead of print. J Leukoc Biol. 2018. PMID: 29603382 Review.
-
MicroRNAs: Fine Tuners of Monocyte Heterogeneity.Front Immunol. 2019 Sep 23;10:2145. doi: 10.3389/fimmu.2019.02145. eCollection 2019. Front Immunol. 2019. PMID: 31608049 Free PMC article. Review.
Cited by
-
Long-Term Recovery of the Adaptive Immune System in Rhesus Macaques After Total Body Irradiation.Adv Radiat Oncol. 2021 Apr 19;6(5):100677. doi: 10.1016/j.adro.2021.100677. eCollection 2021 Sep-Oct. Adv Radiat Oncol. 2021. PMID: 34646962 Free PMC article.
-
Contrasting effects of Western vs Mediterranean diets on monocyte inflammatory gene expression and social behavior in a primate model.Elife. 2021 Aug 2;10:e68293. doi: 10.7554/eLife.68293. Elife. 2021. PMID: 34338633 Free PMC article.
-
Transcriptome of rhesus macaque (Macaca mulatta) exposed to total-body irradiation.Sci Rep. 2021 Mar 18;11(1):6295. doi: 10.1038/s41598-021-85669-6. Sci Rep. 2021. PMID: 33737626 Free PMC article.
-
The NIAID/RNCP Biodosimetry Program: An Overview.Cytogenet Genome Res. 2023;163(3-4):89-102. doi: 10.1159/000534213. Epub 2023 Sep 22. Cytogenet Genome Res. 2023. PMID: 37742625 Free PMC article. Review.
-
Radiation-induced brain injury in non-human primates: A dual tracer PET study with [11C]MPC-6827 and [11C]PiB.Neuroimage Rep. 2025 Mar;5(1):100245. doi: 10.1016/j.ynirp.2025.100245. Epub 2025 Feb 19. Neuroimage Rep. 2025. PMID: 40438303 Free PMC article.
References
-
- Fajardo LF, Stewart JR. Pathogenesis of radiation-induced myocardial fibrosis. Lab Invest 1973; 29:244–57. - PubMed
-
- Cardozo BL, Zoetelief H, van Bekkum DW, Zurcher C, Hagenbeek A. Lung damage following bone marrow transplantation: I. The contribution of irradiation. Int J Radiat Oncol Biol Phys 1985; 11:907–14. - PubMed
-
- Ingold JA, Reed GB, Kaplan HS, Bagshaw MA. Radiation hepatitis. Am J Roentgenol Radium Ther Nucl Med 1965; 93:200–8. - PubMed
-
- Bentzen SM, Thames HD, Overgaard M. Latent-time estimation for late cutaneous and subcutaneous radiation reactions in a single-follow-up clinical study. Radiother Oncol 1989; 15:267–74. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

