Resumption of Immune Checkpoint Inhibitor Therapy After Immune-Mediated Colitis
- PMID: 31163011
- PMCID: PMC6800279
- DOI: 10.1200/JCO.19.00320
Resumption of Immune Checkpoint Inhibitor Therapy After Immune-Mediated Colitis
Abstract
Purpose: Immune checkpoint inhibitor (ICI) therapy often is suspended because of immune-mediated diarrhea and colitis (IMDC). We examined the rate of and risk factors for IMDC recurrence after ICI resumption.
Methods: This retrospective multicenter study examined patients who resumed ICI therapy after improvement of IMDC between January 2010 and November 2018. Univariable and multivariable logistic regression analyses assessed the association of clinical covariates and IMDC recurrence.
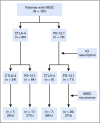
Results: Of the 167 patients in our analysis, 32 resumed an anti-cytotoxic T-cell lymphocyte-4 (CTLA-4) agent, and 135 an anti-programmed cell death 1 or ligand 1 (PD-1/L1) agent. The median age was 60 years (interquartile range [IQR], 50-69 years). The median duration from IMDC to restart of ICI treatment was 49 days (IQR, 23-136 days). IMDC recurred in 57 patients (34%) overall (44% of those receiving an anti-CTLA-4 and 32% of those receiving an anti-PD-1/L1); 47 of these patients (82%) required immunosuppressive therapy for recurrent IMDC, and all required permanent discontinuation of ICI therapy. The median duration from ICI resumption to IMDC recurrence was 53 days (IQR, 22-138 days). On multivariable logistic regression, patients who received anti-PD-1/L1 therapy at initial IMDC had a higher risk of IMDC recurrence (odds ratio [OR], 3.45; 95% CI, 1.59 to 7.69; P = .002). Risk of IMDC recurrence was higher for patients who required immunosuppression for initial IMDC (OR, 3.22; 95% CI, 1.08 to 9.62; P = .019) or had a longer duration of IMDC symptoms in the initial episode (OR, 1.01; 95% CI, 1.00 to 1.03; P = .031). Risk of IMDC recurrence was lower after resumption of anti-PD-1/L1 therapy than after resumption of anti-CTLA-4 therapy (OR, 0.30; 95% CI, 0.11 to 0.81; P = .019).
Conclusion: One third of patients who resumed ICI treatment after IMDC experienced recurrent IMDC. Recurrence of IMDC was less frequent after resumption of anti-PD-1/L1 than after resumption of anti-CTLA-4.
Figures
Comment in
-
Reply to J. Delyon et al.J Clin Oncol. 2019 Dec 20;37(36):3564-3565. doi: 10.1200/JCO.19.02133. Epub 2019 Oct 9. J Clin Oncol. 2019. PMID: 31596634 No abstract available.
-
Recurrence of Immune-Mediated Colitis Upon Immune Checkpoint Inhibitor Resumption: Does Time Matter?J Clin Oncol. 2019 Dec 20;37(36):3563-3564. doi: 10.1200/JCO.19.01891. Epub 2019 Oct 9. J Clin Oncol. 2019. PMID: 31596635 No abstract available.
References
-
- Hargadon KM, Johnson CE, Williams CJ. Immune checkpoint blockade therapy for cancer: An overview of FDA-approved immune checkpoint inhibitors. Int Immunopharmacol. 2018;62:29–39. - PubMed
-
- Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378:158–168. - PubMed
-
- Abu-Sbeih H, Tang T, Ali FS, et al. The impact of immune checkpoint inhibitor-related adverse events and their immunosuppressive treatment on patients’ outcomes. J Immunother Precis Oncol. 2018;1:7–18.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources