Novel Green Biomimetic Approach for Synthesis of ZnO-Ag Nanocomposite; Antimicrobial Activity against Food-borne Pathogen, Biocompatibility and Solar Photocatalysis
- PMID: 31165752
- PMCID: PMC6549174
- DOI: 10.1038/s41598-019-44309-w
Novel Green Biomimetic Approach for Synthesis of ZnO-Ag Nanocomposite; Antimicrobial Activity against Food-borne Pathogen, Biocompatibility and Solar Photocatalysis
Abstract
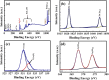
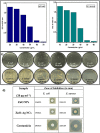
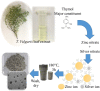
A simple, eco-friendly, and biomimetic approach using Thymus vulgaris (T. vulgaris) leaf extract was developed for the formation of ZnO-Ag nanocomposites (NCs) without employing any stabilizer and a chemical surfactant. T. vulgaris leaf extract was used for the first time, in a novel approach, for green fabrication of ZnO-Ag NCs as a size based reducing agent via the hydrothermal method in a single step. Presence of phenols in T. vulgaris leaf extract has served as both reducing and capping agents that play a critical role in the production of ZnO-Ag NCs. The effect of silver nitrate concentration in the formation of ZnO-Ag NCs was studied. The in-vitro Antimicrobial activity of NCs displayed high antimicrobial potency on selective gram negative and positive foodborne pathogens. Antioxidant activity of ZnO-Ag NCs was evaluated via (2,2-diphenyl-1-picrylhydrazyl) DPPH method. Photocatalytic performance of ZnO-Ag NCs was appraised by degradation of phenol under natural sunlight, which exhibited efficient photocatalytic activity on phenol. Cytotoxicity of the NCs was evaluated using the haemolysis assay. Results of this study reveal that T. vulgaris leaf extract, containing phytochemicals, possess reducing property for ZnO-Ag NCs fabrication and the obtained ZnO-Ag NCs could be employed effectively for biological applications in food science. Therefore, the present study offers a promising way to achieve high-efficiency photocatalysis based on the hybrid structure of semiconductor/metal.
Conflict of interest statement
The authors declare no competing interests.
Figures








References
-
- Khan, I., Saeed, K. & Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem (2017).
-
- Sahay R, Reddy VJ, Ramakrishna S. Synthesis and applications of multifunctional composite nanomaterials. 2014;9:25.
-
- Song JY, Jang H-K, Kim BS. Biological synthesis of gold nanoparticles using Magnolia kobus and Diopyros kaki leaf extracts. Process Biochem. 2009;44:1133–1138. doi: 10.1016/j.procbio.2009.06.005. - DOI
-
- Gallo A, Bianco C, Tosco T, Tiraferri A, Sethi R. Synthesis of eco-compatible bimetallic silver/iron nanoparticles for water remediation and reactivity assessment on bromophenol blue. J. Clean. Prod. 2019;211:1367–1374. doi: 10.1016/j.jclepro.2018.10.298. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

