Pharmacological characterization of the calcium influx pathways involved in nitric oxide production by endothelial cells
- PMID: 31166411
- PMCID: PMC6550436
- DOI: 10.31744/einstein_journal/2019AO4600
Pharmacological characterization of the calcium influx pathways involved in nitric oxide production by endothelial cells
Abstract
Objective: To characterize the calcium influx pathways implicated in the sustained elevation of endothelial intracellular calcium concentration, required for the synthesis and release of relaxing factors.
Methods: We evaluated the effect of the newly synthesized pyrazole derivatives, described as selective inhibitors for ORAI (BTP2/Pyr2 and Pyr6) and TRPC3 (Pyr3 and Pyr10) channels, upon endothelium- and extracellular calcium-dependent relaxations stimulated by acetylcholine and thapsigargin, in pre-constricted rat thoracic aortic rings.
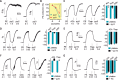
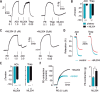
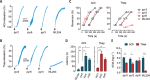
Results: Acetylcholine and thapsigargin responses were completely reverted by Pyr2 and Pyr6 (1 to 3μM). Pyr3 (0.3 to 3μM) caused a rapid reversal of acetylcholine (6.2±0.08mg.s-1) and thapsigargin (3.9±0.25mg.s-1) relaxations, whereas the more selective TRPC3 blocker Pyr10 (1 to 3μM) had no effect. The recently described TRPC4/5 selective blocker, ML204 (1 to 3μM), reverted completely acetylcholine relaxations, but minimally thapsigargin induced ones. Noteworthy, relaxations elicited by GSK1016790A (TRPV4 agonist) were unaffected by pyrazole compounds or ML204. After Pyr2 and Pyr6 pre-incubation, acetylcholine and thapsigargin evoked transient relaxations similar in magnitude and kinetics to those observed in the absence of extracellular calcium. Sodium nitroprusside relaxations as well as phenylephrine-induced contractions (denuded aorta) were not affected by any of pyrazole compounds (1 to 3μM).
Conclusion: These observations revealed a previously unrecognized complexity in rat aorta endothelial calcium influx pathways, which result in production and release of nitric oxide. Pharmacologically distinguishable pathways mediate acetylcholine (ORAI/TRPC other than TRPC3/TRPC4 calcium-permeable channels) and thapsigargin (TRPC4 not required) induced calcium influx.
Objetivo:: Caracterizar as vias do influxo de cálcio envolvidas no aumento sustentado da concentração intracelular de cálcio na célula endotelial, essencial para a síntese e a liberação de fatores relaxantes.
Métodos:: Analisamos o efeito de derivados pirazólicos sintetizados recentemente, descritos como inibidores seletivos para canais ORAI (BTP2/Pyr2 e Pyr6) e TRPC3 (Pyr3 e Pyr10), nos relaxamentos dependentes de endotélio e cálcio extracelular, produzidos por acetilcolina e tapsigargina, em anéis pré-contraídos da aorta torácica de rato.
Resultados:: As respostas de acetilcolina e tapsigargina foram completamente revertidas por Pyr2 e Pyr6 (1 a 3μM). Pyr3 (0,3 a 3μM) produziu reversão rápida dos relaxamentos de acetilcolina (6,2±0,08mg.s−1) e tapsigargina (3,9±0,25mg.s−1), enquanto o bloqueador mais seletivo para TRPC3, Pyr10 (1 a 3μM), não apresentou efeito. ML204 (1 a 3μM), bloqueador seletivo de TRPC4, descrito há pouco tempo, reverteu os relaxamentos induzidos por acetilcolina de forma completa, mas afetou minimamente aqueles produzidos por tapsigargina. Os derivados pirazólicos ou ML204 não afetaram os relaxamentos estimulados com GSK1016790A (TRPV4-agonista). Ainda, após pré-incubação com Pyr2 e Pyr6, acetilcolina e tapsigargina provocaram relaxamentos transitórios semelhantes em magnitude e cinética àqueles observados na ausência de cálcio extracelular. Os relaxamentos do nitroprussiato de sódio e as contrações induzidas pela fenilefrina (aorta sem endotélio) não foram afetados pelos compostos pirazólicos (1 a 3μM).
Conclusão:: Essas observações revelaram uma complexidade desconhecida das vias de influxo de cálcio no endotélio da aorta de rato, que resultam na produção e na liberação de óxido nítrico. Vias distinguíveis farmacologicamente medeiam o influxo estimulado por acetilcolina (ORAI TRPC, diferentes de TRPC3 TRPC4) e tapsigargina (TRPC4 não requerido).
Conflict of interest statement
none.
Figures



Similar articles
-
TRPV4 channel activation leads to endothelium-dependent relaxation mediated by nitric oxide and endothelium-derived hyperpolarizing factor in rat pulmonary artery.Pharmacol Res. 2013 Dec;78:18-27. doi: 10.1016/j.phrs.2013.09.005. Epub 2013 Sep 25. Pharmacol Res. 2013. PMID: 24075884
-
Heteromeric TRPV4/TRPC1 channels mediate calcium-sensing receptor-induced nitric oxide production and vasorelaxation in rabbit mesenteric arteries.Vascul Pharmacol. 2017 Sep;96-98:53-62. doi: 10.1016/j.vph.2017.08.005. Epub 2017 Sep 1. Vascul Pharmacol. 2017. PMID: 28867591 Free PMC article.
-
Central role of intracellular calcium stores in acute flow- and agonist-evoked endothelial nitric oxide release.Br J Pharmacol. 1997 Sep;122(1):117-25. doi: 10.1038/sj.bjp.0701340. Br J Pharmacol. 1997. PMID: 9298537 Free PMC article.
-
Heteromeric TRPV4/TRPC1 channels mediate calcium-sensing receptor-induced relaxations and nitric oxide production in mesenteric arteries: comparative study using wild-type and TRPC1-/- mice.Channels (Austin). 2019 Dec;13(1):410-423. doi: 10.1080/19336950.2019.1673131. Epub 2019 Oct 11. Channels (Austin). 2019. PMID: 31603369 Free PMC article.
-
Novel Chemical Inhibitor of TRPC4 Channels.2010 Dec 15 [updated 2011 May 26]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. 2010 Dec 15 [updated 2011 May 26]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. PMID: 22049577 Free Books & Documents. Review.
Cited by
-
Transient Receptor Potential Canonical (TRPC) Channels: Then and Now.Cells. 2020 Aug 28;9(9):1983. doi: 10.3390/cells9091983. Cells. 2020. PMID: 32872338 Free PMC article. Review.
-
Cracking the Endothelial Calcium (Ca2+) Code: A Matter of Timing and Spacing.Int J Mol Sci. 2023 Nov 26;24(23):16765. doi: 10.3390/ijms242316765. Int J Mol Sci. 2023. PMID: 38069089 Free PMC article. Review.
-
Rap1A Modulates Store-Operated Calcium Entry in the Lung Endothelium: A Novel Mechanism Controlling NFAT-Mediated Vascular Inflammation and Permeability.Arterioscler Thromb Vasc Biol. 2024 Nov;44(11):2271-2287. doi: 10.1161/ATVBAHA.124.321458. Epub 2024 Sep 26. Arterioscler Thromb Vasc Biol. 2024. PMID: 39324266 Free PMC article.
-
Increased transient receptor potential canonical 3 activity is involved in the pathogenesis of detrusor overactivity by dynamic interaction with Na+/Ca2+ exchanger 1.Lab Invest. 2022 Jan;102(1):48-56. doi: 10.1038/s41374-021-00665-8. Epub 2021 Sep 8. Lab Invest. 2022. PMID: 34497367
References
-
- Furchgott RF. Role of endothelium in responses of vascular smooth muscle. Circ Res. 1983;53(5):557–573. Review. - PubMed
-
- Graier WF, Sturek M, Kukovetz WR. Ca2+-Regulation and Endothelial Vascular Function. Endothelium. 1994;1(4):223–236.
-
- Iouzalen L, Lantoine F, Pernollet MG, Millanvoye-Van Brussel E, Devynck MA, David-Dufilho M. SK&F96365 inhibits intracellular Ca2+ pumps and raises cytosolic Ca2+ concentration without production of nitric oxide and von Willebrand factor. Cell Calcium. 1996;20(6):501–508. - PubMed
-
- Korenaga R, Ando J, Ohtsuka A, Sakuma I, Yang W, Toyo-oka T, et al. Close correlation between cytoplasmic Ca++ levels and release of an endothelium-derived relaxing factor from cultured endothelial cells. Cell Struct Funct. 1993;18(2):95–104. - PubMed
-
- Wang Y, Shin WS, Kawaguchi H, Inukai M, Kato M, Sakamoto A, et al. Contribution of sustained Ca2+ elevation for nitric oxide production in endothelial cells and subsequent modulation of Ca2+ transient in vascular smooth muscle cells in coculture. J Biol Chem. 1996;271(10):5647–5655. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

