Molecular Characterization of Monocyte Subsets Reveals Specific and Distinctive Molecular Signatures Associated With Cardiovascular Disease in Rheumatoid Arthritis
- PMID: 31169830
- PMCID: PMC6536567
- DOI: 10.3389/fimmu.2019.01111
Molecular Characterization of Monocyte Subsets Reveals Specific and Distinctive Molecular Signatures Associated With Cardiovascular Disease in Rheumatoid Arthritis
Abstract
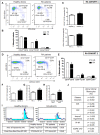
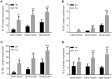
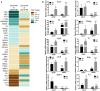
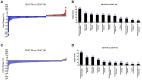
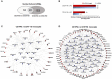
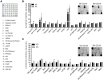
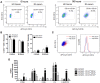
Objectives: This study, developed within the Innovative Medicines Initiative Joint Undertaking project PRECISESADS framework, aimed at functionally characterize the monocyte subsets in RA patients, and analyze their involvement in the increased CV risk associated with RA. Methods: The frequencies of monocyte subpopulations in the peripheral blood of 140 RA patients and 145 healthy donors (HDs) included in the PRECISESADS study were determined by flow cytometry. A second cohort of 50 RA patients and 30 HDs was included, of which CD14+ and CD16+ monocyte subpopulations were isolated using immuno-magnetic selection. Their transcriptomic profiles (mRNA and microRNA), proinflammatory patterns and activated pathways were evaluated and related to clinical features and CV risk. Mechanistic in vitro analyses were further performed. Results: CD14++CD16+ intermediate monocytes were extended in both cohorts of RA patients. Their increased frequency was associated with the positivity for autoantibodies, disease duration, inflammation, endothelial dysfunction and the presence of atheroma plaques, as well as with the CV risk score. CD14+ and CD16+ monocyte subsets showed distinctive and specific mRNA and microRNA profiles, along with specific intracellular signaling activation, indicating different functionalities. Moreover, that specific molecular profiles were interrelated and associated to atherosclerosis development and increased CV risk in RA patients. In vitro, RA serum promoted differentiation of CD14+CD16- to CD14++CD16+ monocytes. Co-culture with RA-isolated monocyte subsets induced differential activation of endothelial cells. Conclusions: Our overall data suggest that the generation of inflammatory monocytes is associated to the autoimmune/inflammatory response that mediates RA. These monocyte subsets, -which display specific and distinctive molecular signatures- might promote endothelial dysfunction and in turn, the progression of atherosclerosis through a finely regulated process driving CVD development in RA.
Keywords: cardiovascular disease; gene profile; microRNAs; monocyte subsets; rheumatoid arthritis.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

