Pyrethroid exposure alters internal and cuticle surface bacterial communities in Anopheles albimanus
- PMID: 31171859
- PMCID: PMC6776023
- DOI: 10.1038/s41396-019-0445-5
Pyrethroid exposure alters internal and cuticle surface bacterial communities in Anopheles albimanus
Abstract
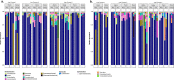
A deeper understanding of the mechanisms underlying insecticide resistance is needed to mitigate its threat to malaria vector control. Following previously identified associations between mosquito microbiota and insecticide resistance, we demonstrate for the first time, the effects of pyrethroid exposure on the microbiota of F1 progeny of field-collected Anopheles albimanus. Larval and adult mosquitoes were exposed to the pyrethroids alphacypermethrin (only adults), permethrin, and deltamethrin. While there were no significant differences in bacterial composition between insecticide-resistant and insecticide-susceptible mosquitoes, bacterial composition between insecticide-exposed and non-exposed mosquitoes was significantly different for alphacypermethrin and permethrin exposure. Along with other bacterial taxa not identified to species, Pantoea agglomerans (a known insecticide-degrading bacterial species) and Pseudomonas fragi were more abundant in insecticide-exposed compared to non-exposed adults, demonstrating that insecticide exposure can alter mosquito bacterial communities. We also show for the first time that the cuticle surfaces of both larval and adult An. albimanus harbor more diverse bacterial communities than their internal microbial niches. Together, these findings demonstrate how insecticide pressure could be selecting for certain bacteria within mosquitoes, especially insecticide-metabolizing bacteria, thus potentially contributing to insecticide resistance.
Conflict of interest statement
The authors declare that they have no conflicts of interest. The findings and conclusions in this paper are those of the authors and do not necessarily represent the official position of the CDC or ASM.
Figures






References
-
- WHO. World Malaria Report 2018. Geneva: World Health Organization; 2018.
-
- WHO. Global report on insecticide resistance in malaria vectors: 2010–2016. Geneva: World Health Organization; 2018.
-
- Quiñones ML, Norris DE, Conn JE, Moreno M, Burkot TR, Bugoro H, et al. Insecticide resistance in areas under investigation by the International Centers of Excellence for Malaria Research: a challenge for malaria control and elimination. Am J Trop Med Hyg. 2015;93:69–78. doi: 10.4269/ajtmh.14-0844. - DOI - PMC - PubMed
-
- Riveron J, Tchouakui M, Mugenzi L, Menze B, Chiang M-C, Wondji C. In Manguin S, Dev V, editors. Insecticide Resistance in Malaria Vectors: An Update at a Global Scale. Towards malaria elimination. A leap forward. IntechOpen; London, UK, 2018.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

