Outcomes and Device Usage for Fully Automated Internet Interventions Designed for a Smartphone or Personal Computer: The MobileQuit Smoking Cessation Randomized Controlled Trial
- PMID: 31172967
- PMCID: PMC6594213
- DOI: 10.2196/13290
Outcomes and Device Usage for Fully Automated Internet Interventions Designed for a Smartphone or Personal Computer: The MobileQuit Smoking Cessation Randomized Controlled Trial
Abstract
Background: Many best practice smoking cessation programs use fully automated internet interventions designed for nonmobile personal computers (desktop computers, laptops, and tablets). A relatively small number of smoking cessation interventions have been designed specifically for mobile devices such as smartphones.
Objective: This study examined the efficacy and usage patterns of two internet-based best practices smoking cessation interventions.
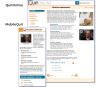
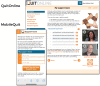
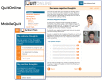
Methods: Overall, 1271 smokers who wanted to quit were randomly assigned to (1) MobileQuit (designed for-and constrained its use to-mobile devices, included text messaging, and embodied tunnel information architecture) or (2) QuitOnline (designed for nonmobile desktop or tablet computers, did not include text messages, and used a flexible hybrid matrix-hierarchical information architecture). Primary outcomes included self-reported 7-day point-prevalence smoking abstinence at 3- and 6-month follow-up assessments. Program visits were unobtrusively assessed (frequency, duration, and device used for access).
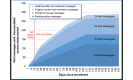
Results: Significantly more MobileQuit participants than QuitOnline participants reported quitting smoking. Abstinence rates using intention-to-treat analysis were 20.7% (131/633) vs 11.4% (73/638) at 3 months, 24.6% (156/633) vs 19.3% (123/638) at 6 months, and 15.8% (100/633) vs 8.8% (56/638) for both 3 and 6 months. Using Complete Cases, MobileQuit's advantage was significant at 3 months (45.6% [131/287] vs 28.4% [73/257]) and the combined 3 and 6 months (40.5% [100/247] vs 25.9% [56/216]) but not at 6 months (43.5% [156/359] vs 34.4% [123/329]). Participants in both conditions reported their program was usable and helpful. MobileQuit participants visited their program 5 times more frequently than did QuitOnline participants. Consistent with the MobileQuit's built-in constraint, 89.46% (8820/9859) of its visits were made on an intended mobile device, whereas 47.72% (691/1448) of visits to QuitOnline used an intended nonmobile device. Among MobileQuit participants, 76.0% (459/604) used only an intended mobile device, 23.0% (139/604) used both mobile and nonmobile devices, and 0.1% (6/604) used only a nonmobile device. Among QuitOnline participants, 31.3% (137/438) used only the intended nonmobile devices, 16.7% (73/438) used both mobile and nonmobile devices, and 52.1% (228/438) used only mobile devices (primarily smartphones).
Conclusions: This study provides evidence for optimizing intervention design for smartphones over a usual care internet approach in which interventions are designed primarily for use on nonmobile devices such as desktop computers, laptops. or tablets. We propose that future internet interventions should be designed for use on all of the devices (multiple screens) that users prefer. We forecast that the approach of designing internet interventions for mobile vs nonmobile devices will be replaced by internet interventions that use a single Web app designed to be responsive (adapt to different screen sizes and operating systems), share user data across devices, embody a pervasive information architecture, and complemented by text message notifications.
Trial registration: ClinicalTrials.gov NCT01952236; https://clinicaltrials.gov/ct2/show/NCT01952236 (Archived by WebCite at http://www.webcitation.org/6zdSxqbf8).
Keywords: device; eHealth; internet; mHealth; smartphone; smoking; tobacco.
©Brian G Danaher, Milagra S Tyler, Ryann C Crowley, Håvar Brendryen, John R Seeley. Originally published in the Journal of Medical Internet Research (http://www.jmir.org), 06.06.2019.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures


References
-
- Brendryen H, Drozd F, Kraft P. A digital smoking cessation program delivered through internet and cell phone without nicotine replacement (happy ending): randomized controlled trial. J Med Internet Res. 2008;10(5):e51. doi: 10.2196/jmir.1005. http://www.jmir.org/2008/5/e51/ v10i5e51 - DOI - PMC - PubMed
-
- Etter J. Comparing the efficacy of two internet-based, computer-tailored smoking cessation programs: a randomized trial. J Med Internet Res. 2005;7(1):e2. doi: 10.2196/jmir.7.1.e2. http://www.jmir.org/2005/1/e2/ v7e2 - DOI - PMC - PubMed
-
- Japuntich SJ, Zehner ME, Smith SS, Jorenby DE, Valdez JA, Fiore MC, Baker TB, Gustafson DH. Smoking cessation via the internet: a randomized clinical trial of an internet intervention as adjuvant treatment in a smoking cessation intervention. Nicotine Tob Res. 2006 Dec;8(Suppl 1):S59–67. - PubMed
-
- McKay HG, Danaher BG, Seeley JR, Lichtenstein E, Gau JM. Comparing two web-based smoking cessation programs: randomized controlled trial. J Med Internet Res. 2008;10(5):e40. doi: 10.2196/jmir.993. http://www.jmir.org/2008/5/e40/ v10i5e40 - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

