Bafilomycin A1 and U18666A Efficiently Impair ZIKV Infection
- PMID: 31174294
- PMCID: PMC6630673
- DOI: 10.3390/v11060524
Bafilomycin A1 and U18666A Efficiently Impair ZIKV Infection
Abstract
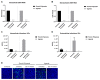
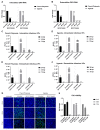
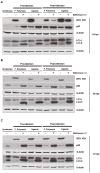
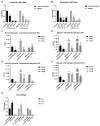
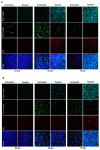
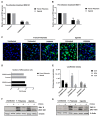
Zika virus (ZIKV) is a highly transmissive virus that belongs to the Flaviviridae family, which comprises several other pathogens that threaten human health. This re-emerging virus gained attention during the outbreak in Brazil in 2016, where a considerable number of microcephaly cases in newborns was associated with ZIKV infection during pregnancy. Lacking a preventive vaccine or antiviral drugs, efforts have been made to better understand the viral life cycle. In light of this, the relevance of the endosomal-lysosomal compartment for the ZIKV life cycle was investigated. A549 and SH-SY5Y cells were infected with either the African strain (associated with mild symptoms) or the French Polynesia strain (associated with neurological complications). For both strains, the V-ATPase inhibitor, bafilomycin A1, efficiently inhibited ZIKV entry and prevented the spread of the infection by interfering with viral maturation. Additionally, affecting cholesterol metabolism and transport with the drug U18666A, which inactivates late endosomes and lysosomes, impairs the viral life cycle. The data presented show a clear antiviral effect of two compounds that target the same compartments in different ways. This highlights the relevance of the endosomal-lysosomal compartment for the viral life cycle that should be considered as a target for antivirals.
Keywords: U18666A; Zika virus; antiviral effect; bafilomycin A1; endosomal–lysosomal compartment; viral life cycle.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Saluzzo J.F., Gonzalez J.P., Hervé J.P., Georges A.J. Enquête sérologique sur la prévalence de certains arborvirus dans la population humaine du sud-est de la République Centrafricaine en 1979. Bull. Soc. Pathol. Exot. Fil. 1981;74:490–499. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

