Hybrid Insulin Peptides Are Autoantigens in Type 1 Diabetes
- PMID: 31175101
- PMCID: PMC6702640
- DOI: 10.2337/db19-0128
Hybrid Insulin Peptides Are Autoantigens in Type 1 Diabetes
Abstract
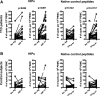
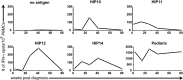
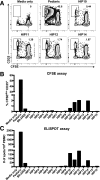
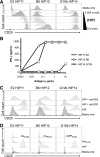
We recently established that hybrid insulin peptides (HIPs) are present in human islets and that T cells reactive to HIPs are found in the residual islets of organ donors with type 1 diabetes (T1D). Here, we investigate whether HIP-reactive T cells are indicative of ongoing autoimmunity in patients with T1D. We used interferon-γ enzyme-linked immune absorbent spot analyses on peripheral blood mononuclear cells (PBMCs) to determine whether patients with new-onset T1D or control subjects displayed T-cell reactivity to a panel of 16 HIPs. We observed that nearly one-half of the patients responded to one or more HIPs. Responses to four HIPs were significantly elevated in patients with T1D but not in control subjects. To characterize the T cells reactive to HIPs, we used a carboxyfluorescein succinimidyl ester-based assay to clone T cells from PBMCs. We isolated six nonredundant, antigen-specific T-cell clones, most of which reacting to their target HIPs in the low nanomolar range. One T-cell clone was isolated from the same patient on two different blood draws, indicating persistence of this T-cell clone in the peripheral blood. This work suggests that HIPs are important target antigens in human subjects with T1D and may play a critical role in disease.
© 2019 by the American Diabetes Association.
Figures

Similar articles
-
Characterization of Human CD4 T Cells Specific for a C-Peptide/C-Peptide Hybrid Insulin Peptide.Front Immunol. 2021 May 25;12:668680. doi: 10.3389/fimmu.2021.668680. eCollection 2021. Front Immunol. 2021. PMID: 34113344 Free PMC article.
-
T-cell responses to hybrid insulin peptides prior to type 1 diabetes development.Proc Natl Acad Sci U S A. 2021 Feb 9;118(6):e2019129118. doi: 10.1073/pnas.2019129118. Proc Natl Acad Sci U S A. 2021. PMID: 33542101 Free PMC article.
-
Identifying New Hybrid Insulin Peptides (HIPs) in Type 1 Diabetes.Front Immunol. 2021 Apr 30;12:667870. doi: 10.3389/fimmu.2021.667870. eCollection 2021. Front Immunol. 2021. PMID: 33995402 Free PMC article. Review.
-
HIPs and HIP-reactive T cells.Clin Exp Immunol. 2019 Dec;198(3):306-313. doi: 10.1111/cei.13335. Epub 2019 Jun 17. Clin Exp Immunol. 2019. PMID: 31132145 Free PMC article. Review.
-
Analysis of self-antigen specificity of islet-infiltrating T cells from human donors with type 1 diabetes.Nat Med. 2016 Dec;22(12):1482-1487. doi: 10.1038/nm.4203. Epub 2016 Oct 31. Nat Med. 2016. PMID: 27798614 Free PMC article.
Cited by
-
Peptide-Spectrum Match Validation with Internal Standards (P-VIS): Internally-Controlled Validation of Mass Spectrometry-Based Peptide Identifications.J Proteome Res. 2021 Jan 1;20(1):236-249. doi: 10.1021/acs.jproteome.0c00355. Epub 2020 Sep 29. J Proteome Res. 2021. PMID: 32924495 Free PMC article.
-
Fifty years of HLA-associated type 1 diabetes risk: history, current knowledge, and future directions.Front Immunol. 2024 Sep 12;15:1457213. doi: 10.3389/fimmu.2024.1457213. eCollection 2024. Front Immunol. 2024. PMID: 39328411 Free PMC article. Review.
-
Why does the immune system destroy pancreatic β-cells but not α-cells in type 1 diabetes?Nat Rev Endocrinol. 2023 Jul;19(7):425-434. doi: 10.1038/s41574-023-00826-3. Epub 2023 Apr 18. Nat Rev Endocrinol. 2023. PMID: 37072614 Review.
-
Tracking DNA-based antigen-specific T cell receptors during progression to type 1 diabetes.Sci Adv. 2023 Dec 8;9(49):eadj6975. doi: 10.1126/sciadv.adj6975. Epub 2023 Dec 8. Sci Adv. 2023. PMID: 38064552 Free PMC article.
-
Dia-B-Ties: B Cells in the Islet-Immune-Cell Interface in T1D.Biomolecules. 2025 Feb 25;15(3):332. doi: 10.3390/biom15030332. Biomolecules. 2025. PMID: 40149868 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

