FGF Signaling Directs the Cell Fate Switch from Neurons to Astrocytes in the Developing Mouse Cerebral Cortex
- PMID: 31175212
- PMCID: PMC6668205
- DOI: 10.1523/JNEUROSCI.2195-18.2019
FGF Signaling Directs the Cell Fate Switch from Neurons to Astrocytes in the Developing Mouse Cerebral Cortex
Abstract
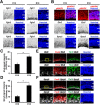
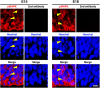
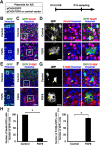
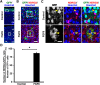
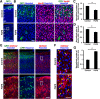
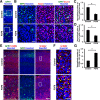
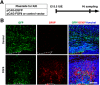
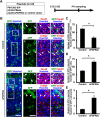
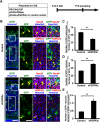
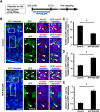
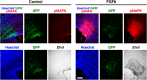
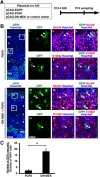
During mammalian neocortical development, neural precursor cells generate neurons first and astrocytes later. The cell fate switch from neurons to astrocytes is a key process generating proper numbers of neurons and astrocytes. Although the intracellular mechanisms regulating this cell fate switch have been well characterized, extracellular regulators are still largely unknown. Here, we uncovered that fibroblast growth factor (FGF) regulates the cell fate switch from neurons to astrocytes in the developing cerebral cortex using mice of both sexes. We found that the FGF signaling pathway is activated in radial glial cells of the ventricular zone at time points corresponding to the switch in cell fate. Our loss- and gain-of-function studies using in utero electroporation indicate that activation of FGF signaling is necessary and sufficient to change cell fates from neurons to astrocytes. We further found that the FGF-induced neuron-astrocyte cell fate switch is mediated by the MAPK pathway. These results indicate that FGF is a critical extracellular regulator of the cell fate switch from neurons to astrocytes in the mammalian cerebral cortex.SIGNIFICANCE STATEMENT Although the intracellular mechanisms regulating the neuron-astrocyte cell fate switch in the mammalian cerebral cortex during development have been well studied, their upstream extracellular regulators remain unknown. By using in utero electroporation, our study provides in vivo data showing that activation of FGF signaling is necessary and sufficient for changing cell fates from neurons to astrocytes. Manipulation of FGF signaling activity led to drastic changes in the numbers of neurons and astrocytes. These results indicate that FGF is a key extracellular regulator determining the numbers of neurons and astrocytes in the mammalian cerebral cortex, and is indispensable for the establishment of appropriate neural circuitry.
Keywords: FGF; astrocyte; cell fate switch; cerebral cortex; neuron.
Copyright © 2019 the authors.
Figures
Similar articles
-
Activation of MAPK/PI3K/SMAD pathways by TGF-β(1) controls differentiation of radial glia into astrocytes in vitro.Dev Neurosci. 2012;34(1):68-81. doi: 10.1159/000338108. Epub 2012 May 25. Dev Neurosci. 2012. PMID: 22652705
-
Caspr Controls the Temporal Specification of Neural Progenitor Cells through Notch Signaling in the Developing Mouse Cerebral Cortex.Cereb Cortex. 2017 Feb 1;27(2):1369-1385. doi: 10.1093/cercor/bhv318. Cereb Cortex. 2017. PMID: 26740489
-
CCAAT/enhancer-binding protein phosphorylation biases cortical precursors to generate neurons rather than astrocytes in vivo.J Neurosci. 2005 Nov 16;25(46):10747-58. doi: 10.1523/JNEUROSCI.2662-05.2005. J Neurosci. 2005. PMID: 16291948 Free PMC article.
-
Radial glial cells as neuronal precursors: a new perspective on the correlation of morphology and lineage restriction in the developing cerebral cortex of mice.Brain Res Bull. 2002 Apr;57(6):777-88. doi: 10.1016/s0361-9230(01)00777-8. Brain Res Bull. 2002. PMID: 12031274 Review.
-
Mechanisms regulating lineage diversity during mammalian cerebral cortical neurogenesis and gliogenesis.Results Probl Cell Differ. 2002;39:27-52. doi: 10.1007/978-3-540-46006-0_2. Results Probl Cell Differ. 2002. PMID: 12357985 Review.
Cited by
-
Evolutionarily conserved concepts in glial cell biology.Curr Opin Neurobiol. 2023 Feb;78:102669. doi: 10.1016/j.conb.2022.102669. Epub 2022 Dec 27. Curr Opin Neurobiol. 2023. PMID: 36577179 Free PMC article. Review.
-
Neural stem/precursor cells dynamically change their epigenetic landscape to differentially respond to BMP signaling for fate switching during brain development.Genes Dev. 2021 Nov 1;35(21-22):1431-1444. doi: 10.1101/gad.348797.121. Epub 2021 Oct 21. Genes Dev. 2021. PMID: 34675062 Free PMC article.
-
Glial cell type-specific gene expression in the mouse cerebrum using the piggyBac system and in utero electroporation.Sci Rep. 2021 Mar 1;11(1):4864. doi: 10.1038/s41598-021-84210-z. Sci Rep. 2021. PMID: 33649472 Free PMC article.
-
The heterogeneity of astrocytes in glaucoma.Front Neuroanat. 2022 Nov 17;16:995369. doi: 10.3389/fnana.2022.995369. eCollection 2022. Front Neuroanat. 2022. PMID: 36466782 Free PMC article. Review.
-
Cyclin-dependent kinase inhibitor p18 regulates lineage transitions of excitatory neurons, astrocytes, and interneurons in the mouse cortex.EMBO J. 2025 Jan;44(2):382-412. doi: 10.1038/s44318-024-00325-9. Epub 2024 Dec 12. EMBO J. 2025. PMID: 39668249 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
