Karyopherin Alpha 2-Expressing Pancreatic Duct Glands and Intra-Islet Ducts in Aged Diabetic C414A-Mutant-CRY1 Transgenic Mice
- PMID: 31179341
- PMCID: PMC6507265
- DOI: 10.1155/2019/7234549
Karyopherin Alpha 2-Expressing Pancreatic Duct Glands and Intra-Islet Ducts in Aged Diabetic C414A-Mutant-CRY1 Transgenic Mice
Abstract
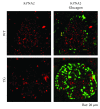
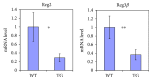
Our earlier studies demonstrated that cysteine414- (zinc-binding site of mCRY1-) alanine mutant mCRY1 transgenic mice (Tg mice) exhibit diabetes characterized by the reduction of β-cell proliferation and by β-cell dysfunction, presumably caused by senescence-associated secretory phenotype- (SASP-) like characters of islets. Earlier studies also showed that atypical duct-like structures in the pancreas developed age-dependently in Tg mice. Numerous reports have described that karyopherin alpha 2 (KPNA2) is highly expressed in cancers of different kinds. However, details of the expression of KPNA2 in pancreatic ductal atypia and in normal pancreatic tissues remain unclear. To assess the feature of the expression of KPNA2 in the development of the ductal atypia and islet architectures, we scrutinized the pancreas of Tg mice histopathologically. Results showed that considerable expression of KPNA2 was observed in pancreatic β-cells, suggesting its importance in maintaining the functions of β-cells. In mature stages, the level of KPNA2 expression was lower in islets of Tg mice than in wild-type controls. At 4 weeks, the expression levels of KPNA2 in islets of Tg mice were the same as those in wild-type controls. These results suggest that the reduction of KPNA2 might contribute to β-cell dysfunction in mature Tg mice. Additionally, the formation of mucin-producing intra-islet ducts, islet fibrosis, and massive T cell recruitment to the islet occurred in aged Tg mice. In exocrine areas, primary pancreatic intraepithelial neoplasias (PanINs) with mucinous pancreatic duct glands (PDGs) emerged in aged Tg mice. High expression of KPNA2 was observed in the ductal atypia. By contrast, KPNA2 expression in normal ducts was quite low. Thus, upregulation of KPNA2 seemed to be correlated with progression of the degree of atypia in pancreatic ductal cells. The SASP-like microenvironment inside islets might play stimulatory roles in the formation of ductal metaplasia inside islets and in islet fibrosis in Tg mice.
Figures






Similar articles
-
Overexpression of heparin-binding EGF-like growth factor in mouse pancreas results in fibrosis and epithelial metaplasia.Gastroenterology. 2003 Apr;124(4):1020-36. doi: 10.1053/gast.2003.50150. Gastroenterology. 2003. PMID: 12671899
-
Unique Aspects of Cryptochrome in Chronobiology and Metabolism, Pancreatic β-Cell Dysfunction, and Regeneration: Research into Cysteine414-Alanine Mutant CRY1.J Diabetes Res. 2016;2016:3459246. doi: 10.1155/2016/3459246. Epub 2016 Dec 26. J Diabetes Res. 2016. PMID: 28105441 Free PMC article. Review.
-
Multipotential nestin-positive stem cells isolated from adult pancreatic islets differentiate ex vivo into pancreatic endocrine, exocrine, and hepatic phenotypes.Diabetes. 2001 Mar;50(3):521-33. doi: 10.2337/diabetes.50.3.521. Diabetes. 2001. PMID: 11246871
-
Karyopherin Alpha 2 Promotes the Inflammatory Response in Rat Pancreatic Acinar Cells Via Facilitating NF-κB Activation.Dig Dis Sci. 2016 Mar;61(3):747-57. doi: 10.1007/s10620-015-3948-6. Epub 2015 Nov 2. Dig Dis Sci. 2016. PMID: 26526450
-
Can pancreatic duct-derived progenitors be a source of islet regeneration?Biochem Biophys Res Commun. 2009 Jun 12;383(4):383-5. doi: 10.1016/j.bbrc.2009.03.114. Epub 2009 Mar 24. Biochem Biophys Res Commun. 2009. PMID: 19324022 Review.
Cited by
-
Increased Nuclear Transporter KPNA2 Contributes to Tumor Immune Evasion by Enhancing PD-L1 Expression in PDAC.J Immunol Res. 2021 Mar 1;2021:6694392. doi: 10.1155/2021/6694392. eCollection 2021. J Immunol Res. 2021. PMID: 33728352 Free PMC article.
-
The RXFP3 receptor is functionally associated with cellular responses to oxidative stress and DNA damage.Aging (Albany NY). 2019 Dec 3;11(23):11268-11313. doi: 10.18632/aging.102528. Epub 2019 Dec 3. Aging (Albany NY). 2019. PMID: 31794429 Free PMC article.
-
IL-6 in inflammation, autoimmunity and cancer.Int Immunol. 2021 Mar 1;33(3):127-148. doi: 10.1093/intimm/dxaa078. Int Immunol. 2021. PMID: 33337480 Free PMC article. Review.
-
Pancreatic Islets and Gestalt Principles.Diabetes. 2020 Sep;69(9):1864-1874. doi: 10.2337/db20-0304. Epub 2020 Jul 15. Diabetes. 2020. PMID: 32669392 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

