Application of Periodontal Ligament-Derived Multipotent Mesenchymal Stromal Cell Sheets for Periodontal Regeneration
- PMID: 31181666
- PMCID: PMC6600219
- DOI: 10.3390/ijms20112796
Application of Periodontal Ligament-Derived Multipotent Mesenchymal Stromal Cell Sheets for Periodontal Regeneration
Abstract
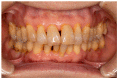
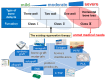
Periodontitis is a chronic inflammatory disorder that causes destruction of the periodontal attachment apparatus including alveolar bone, the periodontal ligament, and cementum. Dental implants have been routinely installed after extraction of periodontitis-affected teeth; however, recent studies have indicated that many dental implants are affected by peri-implantitis, which progresses rapidly because of the failure of the immune system. Therefore, there is a renewed focus on periodontal regeneration aroundnatural teeth. To regenerate periodontal tissue, many researchers and clinicians have attempted to perform periodontal regenerative therapy using materials such as bioresorbable scaffolds, growth factors, and cells. The concept of guided tissue regeneration, by which endogenous periodontal ligament- and alveolar bone-derived cells are preferentially proliferated by barrier membranes, has proved effective, and various kinds of membranes are now commercially available. Clinical studies have shown the significance of barrier membranes for periodontal regeneration; however, the technique is indicated only for relatively small infrabony defects. Cytokine therapies have also been introduced to promote periodontal regeneration, but the indications are also for small size defects. To overcome this limitation, ex vivo expanded multipotent mesenchymal stromal cells (MSCs) have been studied. In particular, periodontal ligament-derived multipotent mesenchymal stromal cells are thought to be a responsible cell source, based on both translational and clinical studies. In this review, responsible cell sources for periodontal regeneration and their clinical applications are summarized. In addition, recent transplantation strategies and perspectives about the cytotherapeutic use of stem cells for periodontal regeneration are discussed.
Keywords: MSCs; clinical study; periodontal ligament; periodontal regeneration; stem cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Allogeneic stem cells from deciduous teeth in treatment for periodontitis in miniature swine.J Periodontol. 2014 Jun;85(6):845-51. doi: 10.1902/jop.2013.130254. Epub 2013 Sep 3. J Periodontol. 2014. PMID: 24001042
-
Regenerative Potential of Bone Morphogenetic Protein 7-Engineered Mesenchymal Stem Cells in Ligature-Induced Periodontitis.Tissue Eng Part A. 2023 Apr;29(7-8):200-210. doi: 10.1089/ten.TEA.2022.0162. Epub 2023 Feb 23. Tissue Eng Part A. 2023. PMID: 36565024
-
Differentiation control of transplanted mesenchymal stem cells (MSCs): a new possible strategy to promote periodontal regeneration.Med Hypotheses. 2008;70(5):944-7. doi: 10.1016/j.mehy.2007.09.013. Epub 2007 Nov 26. Med Hypotheses. 2008. PMID: 18023537
-
Cell sheet engineering and its application for periodontal regeneration.J Tissue Eng Regen Med. 2015 Apr;9(4):343-56. doi: 10.1002/term.1785. Epub 2013 Jul 23. J Tissue Eng Regen Med. 2015. PMID: 23881816 Review.
-
Mesenchymal Stem Cells of Dental Origin for Inducing Tissue Regeneration in Periodontitis: A Mini-Review.Int J Mol Sci. 2018 Mar 22;19(4):944. doi: 10.3390/ijms19040944. Int J Mol Sci. 2018. PMID: 29565801 Free PMC article. Review.
Cited by
-
Polycaprolactone Electrospun Nanofiber Membrane with Sustained Chlorohexidine Release Capability against Oral Pathogens.J Funct Biomater. 2022 Dec 7;13(4):280. doi: 10.3390/jfb13040280. J Funct Biomater. 2022. PMID: 36547540 Free PMC article.
-
Engineering soft-hard tissue interfaces in dental and craniofacial system by spatially controlled bioactivities.Bioact Mater. 2024 Nov 25;45:246-256. doi: 10.1016/j.bioactmat.2024.11.030. eCollection 2025 Mar. Bioact Mater. 2024. PMID: 39659726 Free PMC article. Review.
-
Immunomodulatory Properties of Stem Cells in Periodontitis: Current Status and Future Prospective.Stem Cells Int. 2020 Jul 8;2020:9836518. doi: 10.1155/2020/9836518. eCollection 2020. Stem Cells Int. 2020. PMID: 32724318 Free PMC article. Review.
-
Transcriptome analysis reveals the mechanism of stromal cell-derived factor-1 and exendin-4 synergistically promoted periodontal ligament stem cells osteogenic differentiation.PeerJ. 2021 Aug 27;9:e12091. doi: 10.7717/peerj.12091. eCollection 2021. PeerJ. 2021. PMID: 34532163 Free PMC article.
-
Diagnostic and therapeutic potential of oral cavity-derived exosomes in oral and maxillofacial tissue engineering: current advances and future perspectives.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jun 10. doi: 10.1007/s00210-025-04316-3. Online ahead of print. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40490525 Review.
References
-
- Hegedus Z. The rebuilding of the alveolar processes by bone transplantation. Dent. Cosmos. 1923;65:736–742.
-
- Dominici M., Le Blanc K., Mueller I., Slaper-Cortenbach I., Marini F., Krause D., Deans R., Keating A., Prockop D., Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

