Agrobacterium-mediated vacuum infiltration and floral dip transformation of rapid-cycling Brassica rapa
- PMID: 31182023
- PMCID: PMC6558690
- DOI: 10.1186/s12870-019-1843-6
Agrobacterium-mediated vacuum infiltration and floral dip transformation of rapid-cycling Brassica rapa
Abstract
Background: Rapid-cycling Brassica rapa (RCBr), also known as Wisconsin Fast Plants, are small robust plants with a short lifecycle that are widely used in biology teaching. RCBr have been used for decades but there are no published reports of RCBr genetic transformation. Agrobacterium-mediated vacuum infiltration has been used to transform pakchoi (Brassica rapa ssp. chinensis) and may be suitable for RCBr transformation. The floral dip transformation method, an improved version of vacuum infiltration, could make the procedure easier.
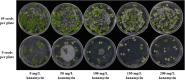
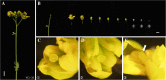
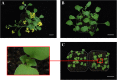
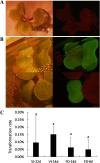
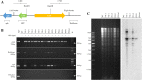
Results: Based on previous findings from Arabidopsis and pakchoi, plants of three different ages were inoculated with Agrobacterium. Kanamycin selection was suboptimal with RCBr; a GFP screen was used to identify candidate transformants. RCBr floral bud dissection showed that only buds with a diameter less than 1 mm carried unsealed carpels, a key point of successful floral dip transformation. Plants across a wide range of inflorescence maturities but containing these immature buds were successfully transformed, at an overall rate of 0.1% (one per 1000 T1 seeds). Transformation was successful using either vacuum infiltration or the floral dip method, as confirmed by PCR and Southern blot.
Conclusion: A genetic transformation system for RCBr was established in this study. This will promote development of new biology teaching tools as well as basic biology research on Brassica rapa.
Keywords: Agrobacterium-mediated transformation; Floral dip; Rapid-cycling Brassica rapa; Vacuum infiltration; Wisconsin fast plants.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
An intensive understanding of vacuum infiltration transformation of pakchoi (Brassica rapa ssp. chinensis).Plant Cell Rep. 2008 Aug;27(8):1369-76. doi: 10.1007/s00299-008-0564-3. Epub 2008 Jun 10. Plant Cell Rep. 2008. PMID: 18542966
-
Floral spray transformation can efficiently generate Arabidopsis transgenic plants.Transgenic Res. 2000 Dec;9(6):471-6. doi: 10.1023/a:1026522104478. Transgenic Res. 2000. PMID: 11206976
-
Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana.Plant J. 1998 Dec;16(6):735-43. doi: 10.1046/j.1365-313x.1998.00343.x. Plant J. 1998. PMID: 10069079
-
Realizing the potential of rapid-cycling Brassica as a model system for use in plant biology research.J Plant Growth Regul. 2000 Sep;19(3):314-25. doi: 10.1007/s003440000036. J Plant Growth Regul. 2000. PMID: 11806422 Review.
-
Arabidopsis thaliana floral dip transformation method.Methods Mol Biol. 2006;343:87-103. doi: 10.1385/1-59745-130-4:87. Methods Mol Biol. 2006. PMID: 16988336 Review.
Cited by
-
A simple and efficient Agrobacterium-mediated in planta transformation protocol for horse gram (Macrotyloma uniflorum Lam. Verdc.).J Genet Eng Biotechnol. 2020 Mar 24;18(1):9. doi: 10.1186/s43141-020-00023-z. J Genet Eng Biotechnol. 2020. PMID: 32206908 Free PMC article.
-
Non-tissue culture genetic modifications for plant improvement.Plant Mol Biol. 2025 May 16;115(3):67. doi: 10.1007/s11103-025-01594-6. Plant Mol Biol. 2025. PMID: 40377725 Review.
-
Optimization of Regeneration and Agrobacterium-Mediated Transformation Protocols for Bi and Multilocular Varieties of Brassica rapa.Plants (Basel). 2022 Dec 29;12(1):161. doi: 10.3390/plants12010161. Plants (Basel). 2022. PMID: 36616290 Free PMC article.
-
Plant genetic transformation: achievements, current status and future prospects.Plant Biotechnol J. 2025 Jun;23(6):2034-2058. doi: 10.1111/pbi.70028. Epub 2025 Mar 7. Plant Biotechnol J. 2025. PMID: 40052992 Free PMC article. Review.
-
Lost in translation: What we have learned from attributes that do not translate from Arabidopsis to other plants.Plant Cell. 2025 May 9;37(5):koaf036. doi: 10.1093/plcell/koaf036. Plant Cell. 2025. PMID: 40371945 Free PMC article. Review.
References
-
- Wisconsin Fast Plants of the University of Wisconsin. https://fastplants.org/. Accessed 3 Dec 2018.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

