Ultrastructural evidence of a mechanosensory function of scale organs (sensilla) in sea snakes (Hydrophiinae)
- PMID: 31183131
- PMCID: PMC6502359
- DOI: 10.1098/rsos.182022
Ultrastructural evidence of a mechanosensory function of scale organs (sensilla) in sea snakes (Hydrophiinae)
Abstract
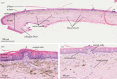
The evolution of epidermal scales was a major innovation in lepidosaurs, providing a barrier to dehydration and physical stress, while functioning as a sensitive interface for detecting mechanical stimuli in the environment. In snakes, mechanoreception involves tiny scale organs (sensilla) that are concentrated on the surface of the head. The fully marine sea snakes (Hydrophiinae) are closely related to terrestrial hydrophiine snakes but have substantially more protruding (dome-shaped) scale organs that often cover a larger portion of the scale surface. Various divergent selection pressures in the marine environment could account for this morphological variation relating to detection of mechanical stimuli from direct contact with stimuli and/or indirect contact via water motion (i.e. 'hydrodynamic reception'), or co-option for alternate sensory or non-sensory functions. We addressed these hypotheses using immunohistochemistry, and light and electron microscopy, to describe the cells and nerve connections underlying scale organs in two sea snakes, Aipysurus laevis and Hydrophis stokesii. Our results show ultrastructural features in the cephalic scale organs of both marine species that closely resemble the mechanosensitive Meissner-like corpuscles that underlie terrestrial snake scale organs. We conclude that the scale organs of marine hydrophiines have retained a mechanosensory function, but future studies are needed to examine whether they are sensitive to hydrodynamic stimuli.
Keywords: cutaneous; mechanoreceptor; scale organs; sea snake; sensilla; skin.
Conflict of interest statement
The authors declare no competing interests.
Figures







References
-
- Lillywhite HB, Maderson PF. 1988. The structure and permeability of integument. Am. Zool. 28, 945–962. (10.1093/icb/28.3.945) - DOI
-
- Maderson PFA, Rabinowitz T, Tandler B, Alibardi L. 1998. Ultrastructural contributions to an understanding of the cellular mechanisms involved in lizard skin shedding with comments on the function and evolution of a unique lepidosaurian phenomenon. J. Morphol. 236, 1–24. (10.1002/(SICI)1097-4687(199804)236:1<1::AID-JMOR1>3.0.CO;2-B) - DOI - PubMed
-
- Dehnhardt G, Mauck B. 2008. The physics and physiology of mechanoreception. In Sensory evolution on the threshold: adaptations in secondarily aquatic vertebrates (eds Nummela S, Thewissen JGM), pp. 287–294 Berkeley, CA: University of California Press.
-
- Povel D, VanDerKooij J. 1997. Scale sensillae of the file snake (Serpentes: Acrochordidae) and some other aquatic and burrowing snakes. Netherlands J. Zool. 47, 443–456.
Associated data
LinkOut - more resources
Full Text Sources
