Cabergoline: a review of its use in the inhibition of lactation for women living with HIV
- PMID: 31183987
- PMCID: PMC6558502
- DOI: 10.1002/jia2.25322
Cabergoline: a review of its use in the inhibition of lactation for women living with HIV
Abstract
Introduction: In developed countries, breastfeeding is not recommended for women living with human immunodeficiency virus (WLWH). However, lactation symptoms can be distressing for women who choose not to breastfeed. There is currently no universal guideline on the most appropriate options for prevention or reduction of lactation symptoms amongst WLWH. This review describes the evidence base for using cabergoline, a dopaminergic agonist, for the post-partum inhibition of lactation for WLWH.
Methods: A scoping review of post-partum pharmaceutical lactation inhibition specific for WLWH was conducted using searches in PubMed, Medline Ovid, EBM Reviews Ovid, Embase, Web of Science and Scopus until 2019. A narrative review of cabergoline pharmacologic properties, therapeutic efficacy, tolerability data and drug interaction data relevant to lactation inhibition was then conducted.
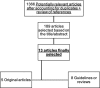
Results and discussion: Among 1366 articles, the scoping review identified 13 relevant publications. Eight guidelines providing guidance regarding lactation inhibition for WLWH and two surveys of medical practice on this topic in UK have been published. Three studies have evaluated the use of pharmaceutical agents in WLWH. Two of these studies evaluated cabergoline and reported it to be an effective method of lactation inhibition in this population. The third study evaluated ethinyl estradiol and bromocriptine use and showed poor efficacy. Cabergoline is a long-acting dopamine D2 agonist and ergot derivative that inhibits prolactin secretion and suppresses physiologic lactation when given as a single oral dose of 1 mg after delivery. Cabergoline is at least as effective as bromocriptine for lactation inhibition with success rates between 78% and 100%. Transient, mild to moderate adverse events to cabergoline are described in clinical trials. Few drug interactions exist as cabergoline is neither a substrate nor an inducer/inhibitor of hepatic cytochrome P450 isoenzymes. There are no reported clinically significant drug-drug interactions between cabergoline and any antiretroviral medications including protease inhibitors.
Conclusions: Cabergoline is a safe and effective pharmacologic option for the prevention of physiological lactation and associated physical symptoms in non-breastfeeding women. Future studies should focus on its safety, efficacy and acceptability among WLWH.
Keywords: HIV; cabergoline; lactation inhibition; lactation suppression; post-partum.
© 2019 The Authors. Journal of the International AIDS Society published by John Wiley & Sons Ltd on behalf of the International AIDS Society.
References
-
- Dunn DT, Newell ML, Ades AE, Peckham CS. Risk of human immunodeficiency virus type 1 transmission through breastfeeding. Lancet. 1992;340(8819):585–8. - PubMed
-
- Loutfy M, Aden M, Wu W, Serghides L. Systematic review of perinatal HIV transmission through breastfeeding for up to twelve months when the mother has viral suppression with combination antiretroviral therapy. 8th International AIDS Society (IAS) Conference on HIV Pathogenesis, Treatment and Prevention. Vancouver, BC, Canada; 2015.
-
- Panel on Treatment of Pregnant Women with HIV Infection and Prevention of Perinatal Transmission . Recommendations for the Use of Antiretroviral Drugs in Pregnant Women with HIV Infection and Interventions to Reduce Perinatal HIV Transmission in the United States. 2018. [cited 2018 Dec 7]. Available from: http://aidsinfo.nih.gov/contentfiles/lvguidelines/PerinatalGL.pdf
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical