Targeting ATGL to rescue BSCL2 lipodystrophy and its associated cardiomyopathy
- PMID: 31185001
- PMCID: PMC6675548
- DOI: 10.1172/jci.insight.129781
Targeting ATGL to rescue BSCL2 lipodystrophy and its associated cardiomyopathy
Abstract
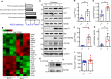
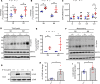
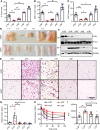
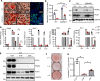
Mutations in BSCL2 gene underlie human type 2 Berardinelli-Seip Congenital Lipodystrophy (BSCL2) disease. Global Bscl2-/- mice recapitulate human BSCL2 lipodystrophy and develop insulin resistance and hypertrophic cardiomyopathy. The pathological mechanisms underlying the development of lipodystrophy and cardiomyopathy in BSCL2 are controversial. Here we report that Bscl2-/- mice develop cardiac hypertrophy due to increased basal IGF1 receptor (IGF1R)-mediated PI3K/AKT signaling. Bscl2-/- hearts exhibited increased adipose triglyceride lipase (ATGL) protein stability and expression causing drastic reduction of glycerolipids. Excessive fatty acid oxidation was overt in Bscl2-/- hearts, partially attributing to the hyperacetylation of cardiac mitochondrial proteins. Intriguingly, pharmacological inhibition or genetic inactivation of ATGL could rescue adipocyte differentiation and lipodystrophy in Bscl2-/- cells and mice. Restoring a small portion of fat mass by ATGL partial deletion in Bscl2-/- mice not only reversed the systemic insulin resistance, but also ameliorated cardiac protein hyperacetylation, normalized cardiac substrate metabolism and improved contractile function. Collectively, our study uncovers novel pathways underlying lipodystrophy-induced cardiac hypertrophy and metabolic remodeling and pinpoints ATGL as a downstream target of BSCL2 in regulating the development of lipodystrophy and its associated cardiomyopathy.
Keywords: Adipose tissue; Cardiology; Heart failure; Metabolism.
Conflict of interest statement
Figures

Similar articles
-
Berardinelli-Seip congenital lipodystrophy 2/SEIPIN determines brown adipose tissue maintenance and thermogenic programing.Mol Metab. 2020 Jun;36:100971. doi: 10.1016/j.molmet.2020.02.014. Epub 2020 Mar 4. Mol Metab. 2020. PMID: 32246911 Free PMC article.
-
Role of Seipin in Human Diseases and Experimental Animal Models.Biomolecules. 2022 Jun 17;12(6):840. doi: 10.3390/biom12060840. Biomolecules. 2022. PMID: 35740965 Free PMC article. Review.
-
BSCL2/Seipin deficiency in hearts causes cardiac energy deficit and dysfunction via inducing excessive lipid catabolism.Clin Transl Med. 2022 Apr;12(4):e736. doi: 10.1002/ctm2.736. Clin Transl Med. 2022. PMID: 35384404 Free PMC article.
-
Berardinelli-Seip congenital lipodystrophy 2 regulates adipocyte lipolysis, browning, and energy balance in adult animals.J Lipid Res. 2015 Oct;56(10):1912-25. doi: 10.1194/jlr.M060244. Epub 2015 Aug 12. J Lipid Res. 2015. PMID: 26269358 Free PMC article.
-
Function of seipin: new insights from Bscl2/seipin knockout mouse models.Biochimie. 2014 Jan;96:166-72. doi: 10.1016/j.biochi.2013.06.022. Epub 2013 Jul 2. Biochimie. 2014. PMID: 23831461 Review.
Cited by
-
Neddylation and Its Target Cullin 3 Are Essential for Adipocyte Differentiation.Cells. 2024 Oct 5;13(19):1654. doi: 10.3390/cells13191654. Cells. 2024. PMID: 39404417 Free PMC article.
-
Berardinelli-Seip congenital lipodystrophy 2/SEIPIN determines brown adipose tissue maintenance and thermogenic programing.Mol Metab. 2020 Jun;36:100971. doi: 10.1016/j.molmet.2020.02.014. Epub 2020 Mar 4. Mol Metab. 2020. PMID: 32246911 Free PMC article.
-
Myocardin reverses insulin resistance and ameliorates cardiomyopathy by increasing IRS-1 expression in a murine model of lipodystrophy caused by adipose deficiency of vacuolar H+-ATPase V0d1 subunit.Theranostics. 2024 Mar 11;14(5):2246-2264. doi: 10.7150/thno.93192. eCollection 2024. Theranostics. 2024. PMID: 38505620 Free PMC article.
-
Role of Seipin in Human Diseases and Experimental Animal Models.Biomolecules. 2022 Jun 17;12(6):840. doi: 10.3390/biom12060840. Biomolecules. 2022. PMID: 35740965 Free PMC article. Review.
-
Lipotoxicity: a driver of heart failure with preserved ejection fraction?Clin Sci (Lond). 2021 Oct 15;135(19):2265-2283. doi: 10.1042/CS20210127. Clin Sci (Lond). 2021. PMID: 34643676 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

