Hyaluronan-based hydrogels as dermal fillers: The biophysical properties that translate into a "volumetric" effect
- PMID: 31185059
- PMCID: PMC6559669
- DOI: 10.1371/journal.pone.0218287
Hyaluronan-based hydrogels as dermal fillers: The biophysical properties that translate into a "volumetric" effect
Abstract
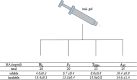
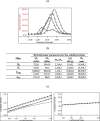
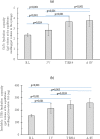
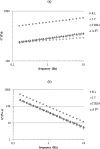
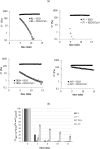
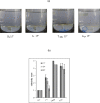
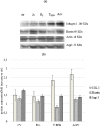
Biophysical and biochemical data on hyaluronan (HA)-based dermal fillers strongly support their optimal use and design to meet specific requisites. Here, four commercially available (in Europe) HA "volumetric" fillers, among the most used in the clinical practice, have been characterized in vitro. Analyses revealed the highest amounts of water-soluble HA reported so far and provided hydrodynamic data for these soluble polymeric fractions. Volumetric gels exhibit a wide range of rigidity with most of them showing G' values around 200-300Pa. They greatly differ in cohesivity. 1mL of gel hydrates up to 2.4-3.2mL. The products completely solubilize due to Bovine Testicular Hyaluronidase (BTH)'s action, thus predicting in vivo complete resorption. For the first time, filler degradation due to reactive oxygen species (ROS) was studied by rheological measurements and a rank in stability was established. Studies using Human Dermal Fibroblasts (HDF) indicated a positive biological response to the HA networks. Further, gel capacity to prompt collagen I, elastin and aquaporin3 synthesis was demonstrated, thus suggesting a positive effect on skin elasticity and hydration, besides the physical volumetric action. The findings are the first wide assessment of features for the volumetric class of HA-fillers and include first data on their resistance to degradation by ROS and biological effects on HDF. The study represents a valuable contribution to the understanding of HA-fillers, useful to optimize their use and manufacture.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Hyaluronan Hydrogels for Injection in Superficial Dermal Layers: An In Vitro Characterization to Compare Performance and Unravel the Scientific Basis of Their Indication.Int J Mol Sci. 2021 Jun 2;22(11):6005. doi: 10.3390/ijms22116005. Int J Mol Sci. 2021. PMID: 34199374 Free PMC article.
-
The reconstructed skin model as a new tool for investigating in vitro dermal fillers: increased fibroblast activity by hyaluronic acid.Eur J Dermatol. 2015 Jul-Aug;25(4):312-22. doi: 10.1684/ejd.2015.2563. Eur J Dermatol. 2015. PMID: 26065380
-
Biophysical and biological characterization of a new line of hyaluronan-based dermal fillers: A scientific rationale to specific clinical indications.Mater Sci Eng C Mater Biol Appl. 2016 Nov 1;68:565-572. doi: 10.1016/j.msec.2016.06.008. Epub 2016 Jun 7. Mater Sci Eng C Mater Biol Appl. 2016. PMID: 27524055
-
The science of hyaluronic acid dermal fillers.J Cosmet Laser Ther. 2008 Mar;10(1):35-42. doi: 10.1080/14764170701774901. J Cosmet Laser Ther. 2008. PMID: 18330796 Review.
-
Hyaluronic Acid Fillers in Soft Tissue Regeneration.Facial Plast Surg. 2017 Feb;33(1):87-96. doi: 10.1055/s-0036-1597685. Epub 2017 Feb 22. Facial Plast Surg. 2017. PMID: 28226376 Review.
Cited by
-
Case report: Improving quality of life through hyaluronic acid-based fillers after orbital cancer treatment.Front Oncol. 2024 Dec 18;14:1501556. doi: 10.3389/fonc.2024.1501556. eCollection 2024. Front Oncol. 2024. PMID: 39744006 Free PMC article.
-
A Scientific Framework for Comparing Hyaluronic Acid Filler Crosslinking Technologies.Gels. 2025 Jun 23;11(7):487. doi: 10.3390/gels11070487. Gels. 2025. PMID: 40710649 Free PMC article.
-
Evaluation of the Rheologic and Physicochemical Properties of a Novel Hyaluronic Acid Filler Range with eXcellent Three-Dimensional Reticulation (XTR™) Technology.Polymers (Basel). 2020 Jul 24;12(8):1644. doi: 10.3390/polym12081644. Polymers (Basel). 2020. PMID: 32722003 Free PMC article.
-
The Intrinsic Relation between the Hydrogel Structure and In Vivo Performance of Hyaluronic Acid Dermal Fillers: A Comparative Study of Four Typical Dermal Fillers.Tissue Eng Regen Med. 2023 Jun;20(3):503-517. doi: 10.1007/s13770-023-00533-0. Epub 2023 Apr 11. Tissue Eng Regen Med. 2023. PMID: 37041433 Free PMC article.
-
Potential of Biofermentative Unsulfated Chondroitin and Hyaluronic Acid in Dermal Repair.Int J Mol Sci. 2022 Feb 1;23(3):1686. doi: 10.3390/ijms23031686. Int J Mol Sci. 2022. PMID: 35163608 Free PMC article.
References
-
- Ave MN, de Almeida Issa M.C. Hyaluronic Acid Dermal Filler: Physical Properties and Its Indications In: Issa M., Tamura B. (eds) Botulinum Toxins, Fillers and Related Substances. Clinical Approaches and Procedures in Cosmetic Dermatology. Springer, Cham; 2018. Vol.4, p. 187–197
-
- Moillard SG, Betemps JB, Hadjab B, Topchian D, Micheels P, Salomon D. Key rheological properties of hyaluronic acid fillers: from tissue integration to product degradation. Plast Aesthet Res. 2018; 5: 17–24.
-
- Basta SL. Cosmetic fillers perspective on the industry. Facial Plast Surg Clin N Am. 2015; 23: 417–21. - PubMed
-
- Cheng LY, Sun XM; Tang MY, Jin R, Cui WG, Zhang YG. An update review on recent skin fillers. Plast Aesthet Res. 2016; 3: 92–99.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

