Rotary mechanical circulatory support systems
- PMID: 31186935
- PMCID: PMC6453075
- DOI: 10.1177/2055668317725994
Rotary mechanical circulatory support systems
Abstract
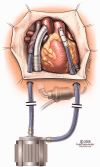
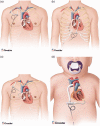
A detailed survey of the current trends and recent advances in rotary mechanical circulatory support systems is presented in this paper. Rather than clinical reports, the focus is on technological aspects of these rehabilitating devices as a reference for engineers and biomedical researchers. Existing trends in flow regimes, flow control, and bearing mechanisms are summarized. System specifications and applications of the most prominent continuous-flow ventricular assistive devices are provided. Based on the flow regime, pumps are categorized as axial flow, centrifugal flow, and mixed flow. Unique characteristics of each system are unveiled through an examination of the structure, bearing mechanism, impeller design, flow rate, and biocompatibility. A discussion on the current limitations is provided to invite more studies and further improvements.
Keywords: Assistive technology; biomedical devices; heart failure; life support systems; mechanical circulatory support; orthotics; rehabilitation devices; ventricular assistive device.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures











Similar articles
-
Axial and centrifugal continuous-flow rotary pumps: a translation from pump mechanics to clinical practice.J Heart Lung Transplant. 2013 Jan;32(1):1-11. doi: 10.1016/j.healun.2012.10.001. J Heart Lung Transplant. 2013. PMID: 23260699 Review.
-
Development of mechanical circulatory support devices in China.Artif Organs. 2009 Nov;33(11):1009-14. doi: 10.1111/j.1525-1594.2009.00913.x. Artif Organs. 2009. PMID: 20021474
-
The Society of Thoracic Surgeons Intermacs database annual report: Evolving indications, outcomes, and scientific partnerships.J Heart Lung Transplant. 2019 Feb;38(2):114-126. doi: 10.1016/j.healun.2018.11.013. J Heart Lung Transplant. 2019. PMID: 30691593
-
Third Annual Report From the ISHLT Mechanically Assisted Circulatory Support Registry: A comparison of centrifugal and axial continuous-flow left ventricular assist devices.J Heart Lung Transplant. 2019 Apr;38(4):352-363. doi: 10.1016/j.healun.2019.02.004. J Heart Lung Transplant. 2019. PMID: 30945637
-
Rotary piston blood pumps: past developments and future potential of a unique pump type.Expert Rev Med Devices. 2016 Aug;13(8):759-71. doi: 10.1080/17434440.2016.1207522. Epub 2016 Jul 11. Expert Rev Med Devices. 2016. PMID: 27353827 Review.
Cited by
-
Left atrial assist device for heart failure with preserved ejection fraction: initial results with torque control mode in diastolic heart failure model.Heart Fail Rev. 2023 Mar;28(2):287-296. doi: 10.1007/s10741-021-10117-6. Epub 2021 May 1. Heart Fail Rev. 2023. PMID: 33931816 Review.
-
Design and Fabrication of Pneumatically Actuated Valveless Pumps.Micromachines (Basel). 2021 Dec 23;13(1):16. doi: 10.3390/mi13010016. Micromachines (Basel). 2021. PMID: 35056181 Free PMC article.
-
Cardiac power output ratio: Novel survival predictor after percutaneous ventricular assist device in cardiogenic shock.ESC Heart Fail. 2024 Dec;11(6):3674-3686. doi: 10.1002/ehf2.14949. Epub 2024 Jul 9. ESC Heart Fail. 2024. PMID: 38982624 Free PMC article.
-
Linking Computational Fluid Dynamics Modeling to Device-Induced Platelet Defects in Mechanically Assisted Circulation.ASAIO J. 2024 Dec 1;70(12):1085-1093. doi: 10.1097/MAT.0000000000002242. Epub 2024 May 19. ASAIO J. 2024. PMID: 38768482
-
Development of Inspired Therapeutics Pediatric VAD: Benchtop Evaluation of Impeller Performance and Torques for MagLev Motor Design.Cardiovasc Eng Technol. 2022 Apr;13(2):307-317. doi: 10.1007/s13239-021-00578-z. Epub 2021 Sep 13. Cardiovasc Eng Technol. 2022. PMID: 34518953 Free PMC article.
References
-
- Agarwal S, High KM. Newer-generation ventricular assist devices. Best Pract Res Clin Anaesthesiol 2012; 26: 117–130. - PubMed
-
- Schumer EM, Black MC, Monreal G, et al. Left ventricular assist devices: current controversies and future directions. Eur Heart J 2016; 37: 3434–3439. - PubMed
-
- Loforte A, Musumeci F, Montalto A, et al. Use of mechanical circulatory support devices in end-stage heart failure patients. J Card Surg 2014; 29: 717–722. - PubMed
-
- Slaughter MS, Bostic R, Tong K, et al. Temporal changes in hospital costs for left ventricular assist device implantation. J Card Surg 2011; 26: 535–541. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

