Integration of PEG 400 into a self-nanoemulsifying drug delivery system improves drug loading capacity and nasal mucosa permeability and prolongs the survival of rats with malignant brain tumors
- PMID: 31190814
- PMCID: PMC6530554
- DOI: 10.2147/IJN.S193617
Integration of PEG 400 into a self-nanoemulsifying drug delivery system improves drug loading capacity and nasal mucosa permeability and prolongs the survival of rats with malignant brain tumors
Abstract
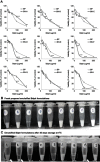
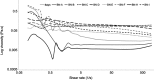
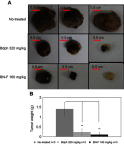
Introduction: Kolliphor® EL (K-EL) is among the most useful surfactants in the preparation of emulsions. However, it is associated with low hydrophobic drug loading in the resulting emulsified formulation. Methods: In this study, a formulation for intranasal administration of butylidenephthalide (Bdph), a candidate drug against glioblastoma (GBM), was prepared. Physical characteristics of the formulation such as particle size, zeta potential, conductivity, and viscosity were assessed, as well as its cytotoxicity and permeability, in order to optimize the formulation and improve its drug loading capacity. Results: The optimized formulation involved the integration of polyethylene glycol 400 (PEG 400) in K-EL to encapsulate Bdph dissolved in dimethyl sulfoxide (DMSO), and it exhibited higher drug loading capacity and drug solubility in water than the old formulation, which did not contain PEG 400. Incorporation of PEG 400 as a co-surfactant increased Bdph loading capacity to up to 50% (v/v), even in formulations using Kolliphor® HS 15 (K-HS15) as a surfactant, which is less compatible with Bdph than K-EL. The optimized Bdph formulation presented 5- and 2.5-fold higher permeability and cytotoxicity, respectively, in human GBM than stock Bdph. This could be attributed to the high drug loading capacity and the high polarity index due to DMSO, which increases the compatibility between the drug and the cell. Rats bearing a brain glioma treated with 160 mg/kg intranasal emulsified Bdph had a mean survival of 37 days, which is the same survival time achieved by treatment with 320 mg/kg stock Bdph. This implies that the optimized emulsified formulation required only half the Bdph dose to achieve an efficacy similar to that of stock Bdph in the treatment of animals with malignant brain tumor.
Keywords: butylidenephthalide; glioblastoma; intranasal administration; loading capacity; permeability; polyethylene glycol 400.
Conflict of interest statement
The formulation has been patented in the USA (Patent No. US9504751 (B2)). Miss Yu-Shuan Chen reports non-financial support from National Science Council of the Republic of China, Taiwan, during the conduct of the study. Dr Shinn-Zong Lin reports non-financial support from National Science Council of the Republic of China, Taiwan, during the conduct of the study. Dr Tzyy-Wen Chiou reports grants from National Science Council, Taiwan, Republic of China, during the conduct of the study. The authors report no further conflicts of interest in this work.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

