Cost-effectiveness analysis of the use of letermovir for the prophylaxis of cytomegalovirus in adult cytomegalovirus seropositive recipients undergoing allogenic hematopoietic stem cell transplantation in Italy
- PMID: 31190905
- PMCID: PMC6512572
- DOI: 10.2147/IDR.S196282
Cost-effectiveness analysis of the use of letermovir for the prophylaxis of cytomegalovirus in adult cytomegalovirus seropositive recipients undergoing allogenic hematopoietic stem cell transplantation in Italy
Abstract
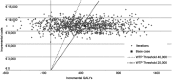
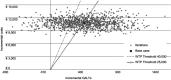
Background: The aim of the analysis is to assess the efficiency of the allocation of economic resources related to the use of letermovir cytomegalovirus (CMV) prophylaxis in adult seropositive recipients (R+) patients receiving an allogenic hematopoietic stem cell transplantation (HSCT), compared with a no-prophylaxis strategy, assuming preemptive antiviral administration in both groups from the perspective of the Italian National Health Service (NHS), through a cost-effectiveness analysis. Methods: The model used is based on a decision tree which simulates on a lifetime horizon the progression of CMV infection, considering two alternatives: the use of letermovir CMV prophylaxis, followed by preemptive therapy in case of clinically significant CMV infection, or the avoided use of letermovir CMV prophylaxis, considering direct medical costs (referred to 2018) and quality-adjusted life years (QALYs), both discounted considering a 3% annual rate. Two scenarios were considered, representing the differences related to regional contexts and clinical practice of different typologies of hospitals (public or private accredited with Regional Health Services). Results: The use of letermovir prophylaxis compared with no prophylaxis strategy would lead to an increase of QALYs and direct medical costs in the two scenarios considered, with a mean increase of 0.45 QALYs, and an increase of direct medical costs of 10,222.4 € and of 10,809.9 € in the two scenarios. The incremental cost-effectiveness ratios are equal to 22,564 €/QALY and 23,861 €/QALY. The probabilistic sensitivity analysis conducted showed a percentage of results below the threshold of 40,000 €/QALY of 67.4% and 71.3%; and below a threshold of 25,000 €/QALY equal to 50.4% and to 53.0%. Conclusions: The use of letermovir CMV prophylaxis in adult R+ patients receiving allogenic HSCT, compared with a no-prophylaxis strategy, would be cost-effective for the Italian NHS considering the incremental cost-effectiveness thresholds of 40,000 €/QALY and of 25,000 €/QALY.
Keywords: allogenic hematopoietic stem cell transplantation; cost-effectiveness analysis; cytomegalovirus; letermovir.
Conflict of interest statement
DC declares advisory board fees from Mundipharma, Takeda and Abbvie. CG has received honoraria from Gilead Sciences, Astellas Pharma, Basilea, MSD, Pfizer Pharmaceuticals, and Celgene. UR report grants from MSD, during the conduct of the study; personal fees from Astrazeneca, personal fees from Bayer, personal fees from Italfarmaco, outside the submitted work. The remaining authors report no conflicts of interest in this work.
Figures
Similar articles
-
Cost-effectiveness of letermovir as cytomegalovirus prophylaxis in adult recipients of allogeneic hematopoietic stem cell transplantation in Hong Kong.J Med Econ. 2020 Dec;23(12):1485-1492. doi: 10.1080/13696998.2020.1843321. Epub 2020 Nov 12. J Med Econ. 2020. PMID: 33155494
-
Cost-effectiveness analysis of cytomegalovirus prophylaxis in allogeneic hematopoietic cell transplant recipients from a US payer perspective.J Med Virol. 2021 Jun;93(6):3786-3794. doi: 10.1002/jmv.26462. Epub 2020 Oct 5. J Med Virol. 2021. PMID: 32844453
-
Real-World Safety and Effectiveness of Letermovir in Patients Undergoing Allogenic Hematopoietic Stem Cell Transplantation: Final Results of Post-Marketing Surveillance in Japan.Clin Drug Investig. 2024 Jul;44(7):527-540. doi: 10.1007/s40261-024-01376-w. Epub 2024 Jun 27. Clin Drug Investig. 2024. PMID: 38935253 Free PMC article.
-
Role of letermovir for prevention of cytomegalovirus infection after allogeneic haematopoietic stem cell transplantation.Curr Opin Infect Dis. 2018 Aug;31(4):286-291. doi: 10.1097/QCO.0000000000000459. Curr Opin Infect Dis. 2018. PMID: 29746444 Review.
-
Letermovir for the prevention of cytomegalovirus infection in adult cytomegalovirus-seropositive hematopoietic stem cell transplant recipients.Expert Rev Clin Pharmacol. 2018 Oct;11(10):931-941. doi: 10.1080/17512433.2018.1500897. Epub 2018 Sep 18. Expert Rev Clin Pharmacol. 2018. PMID: 30004790 Review.
Cited by
-
Cost Effectiveness of Letermovir for Cytomegalovirus Prophylaxis Compared with Pre-Emptive Therapy in Allogeneic Hematopoietic Stem Cell Transplant Recipients in the United States.Pharmacoecon Open. 2023 May;7(3):393-404. doi: 10.1007/s41669-023-00398-y. Epub 2023 Feb 25. Pharmacoecon Open. 2023. PMID: 36840894 Free PMC article.
-
Letermovir Prophylaxis for Cytomegalovirus Infection in Allogeneic Stem Cell Transplantation: A Real-World Experience.Front Oncol. 2021 Sep 6;11:740079. doi: 10.3389/fonc.2021.740079. eCollection 2021. Front Oncol. 2021. PMID: 34616684 Free PMC article.
-
Real-Life Data on the Efficacy and Safety of Letermovir for Primary Prophylaxis of Cytomegalovirus in Allogeneic Hematopoietic Stem Cell Recipients: A Single-Center Analysis.Turk J Haematol. 2024 Mar 1;41(1):9-15. doi: 10.4274/tjh.galenos.2024.2024.0026. Epub 2024 Feb 13. Turk J Haematol. 2024. PMID: 38345092 Free PMC article.
-
Advances in CMV Management: A Single Center Real-Life Experience.Front Cell Dev Biol. 2020 Oct 27;8:534268. doi: 10.3389/fcell.2020.534268. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33195184 Free PMC article.
-
Healthcare Resource Use and Costs of Allogeneic Hematopoietic Stem Cell Transplantation Complications: A Scoping Review.Curr Oncol. 2025 May 16;32(5):283. doi: 10.3390/curroncol32050283. Curr Oncol. 2025. PMID: 40422542 Free PMC article.
References
-
- Rowe RG, Guo D, Lee M, Margossian S, London WB, Lehmann L. Cytomegalovirus infection in pediatric hematopoietic stem cell transplantation: risk factors for primary infection and cases of recurrent and late infection at a single center. Biol Blood Marrow Transplant. 2016;22(7):1275–1283. doi:10.1016/j.bbmt.2016.04.004 - DOI - PMC - PubMed
-
- George B, Pati N, Gilroy N, et al. Pre-transplant cytomegalovirus (CMV) serostatus remains the most important determinant of CMV reactivation after allogeneic hematopoietic stem cell transplantation in the era of surveillance and preemptive therapy. Transpl Infect Dis. 2010;12(4):322–329. doi:10.1111/j.1399-3062.2010.00504.x - DOI - PubMed
-
- Miller W, Flynn P, McCullough J, et al. Cytomegalovirus infection after bone marrow transplantation: an association with acute graft-v-host disease. Blood. 1986;67(4):1162–1167. - PubMed
-
- Ljungman P, Perez-Bercoff L, Jonsson J, et al. Risk factors for the development of cytomegalovirus disease after allogeneic stem cell transplantation. Haematologica. 2006;91(1):78–83. - PubMed
-
- Italian Bone Marrow Transplantation Group. Libro GITMO 2017–2018. Available from: http://www.gitmo.it/index.php/libri-gitmo-pub. Accessed September, 2018.
LinkOut - more resources
Full Text Sources