Electrical Stimulation of the Lateral Entorhinal Cortex Causes a Frequency-Specific BOLD Response Pattern in the Rat Brain
- PMID: 31191231
- PMCID: PMC6547013
- DOI: 10.3389/fnins.2019.00539
Electrical Stimulation of the Lateral Entorhinal Cortex Causes a Frequency-Specific BOLD Response Pattern in the Rat Brain
Abstract
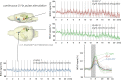
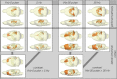
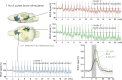
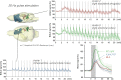
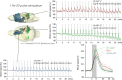
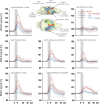
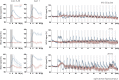
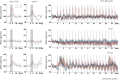
Although deep brain stimulation of the entorhinal cortex has recently shown promise in the treatment of early forms of cognitive decline, the underlying neurophysiological processes remain elusive. Therefore, the lateral entorhinal cortex (LEC) was stimulated with trains of continuous 5 Hz and 20 Hz pulses or with bursts of 100 Hz pulses to visualize activated neuronal networks, i.e., neuronal responses in the dentate gyrus and BOLD responses in the entire brain were simultaneously recorded. Electrical stimulation of the LEC caused a wide spread pattern of BOLD responses. Dependent on the stimulation frequency, BOLD responses were only triggered in the amygdala, infralimbic, prelimbic, and dorsal peduncular cortex (5 Hz), or in the nucleus accumbens, piriform cortex, dorsal medial prefrontal cortex, hippocampus (20 Hz), and contralateral entorhinal cortex (100 Hz). In general, LEC stimulation caused stronger BOLD responses in frontal cortex regions than in the hippocampus. Identical stimulation of the perforant pathway, a fiber bundle projecting from the entorhinal cortex to the dentate gyrus, hippocampus proper, and subiculum, mainly elicited significant BOLD responses in the hippocampus but rarely in frontal cortex regions. Consequently, BOLD responses in frontal cortex regions are mediated by direct projections from the LEC rather than via signal propagation through the hippocampus. Thus, the beneficial effects of deep brain stimulation of the entorhinal cortex on cognitive skills might depend more on an altered prefrontal cortex than hippocampal function.
Keywords: BOLD fMRI; amygdala; in vivo electrophysiology; limbic system; piriform cortex; prefrontal cortex.
Figures


References
-
- Angenstein F., Krautwald K., Wetzel W., Scheich H. (2013). Perforant pathway stimulation as a conditioned stimulus for active avoidance learning triggers BOLD responses in various target regions of the hippocampus: a combined fMRI and electrophysiological study. Neuroimage 75 213–227. 10.1016/j.neuroimage.2013.03.007 - DOI - PubMed
-
- Bovet-Carmona M., Menigoz A., Pinto S., Tambuyzer T., Krautwald K., Voets T., et al. (2018). Disentangling the role of TRPM4 in hippocampus-dependent plasticity and learning: an electrophysiological, behavioral and FMRI approach. Brain Struct. Funct. 223 3557–3576. 10.1007/s00429-018-1706-1 - DOI - PubMed
LinkOut - more resources
Full Text Sources

