Tellurium Compounds Prevent and Reverse Type-1 Diabetes in NOD Mice by Modulating α4β7 Integrin Activity, IL-1β, and T Regulatory Cells
- PMID: 31191514
- PMCID: PMC6549385
- DOI: 10.3389/fimmu.2019.00979
Tellurium Compounds Prevent and Reverse Type-1 Diabetes in NOD Mice by Modulating α4β7 Integrin Activity, IL-1β, and T Regulatory Cells
Abstract
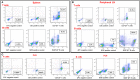
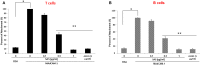
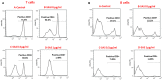
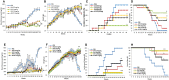
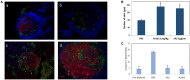
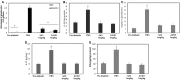
The study shows that treatment of NOD mice with either of two tellurium-based small molecules, AS101 [ammonium trichloro(dioxoethylene-o,o')tellurate] or SAS [octa-O-bis-(R,R)-tartarate ditellurane] could preserve β cells function and mass. These beneficial effects were reflected in decreased incidence of diabetes, improved glucose clearance, preservation of body weight, and increased survival. The normal glucose levels were associated with increased insulin levels, preservation of β cell mass and increased islet size. Importantly, this protective activity could be demonstrated when the compounds were administered either at the early pre-diabetic phase with no or initial insulitis, at the pre-diabetic stage with advanced insulitis, or even at the advanced, overtly diabetic stage. We further demonstrate that both tellurium compounds prevent migration of autoimmune lymphocytes to the pancreas, via inhibition of the α4β7 integrin activity. Indeed, the decreased migration resulted in diminished pancreatic islets damage both with respect to their size, β cell function, and caspase-3 activity, the hallmark of apoptosis. Most importantly, AS101 and SAS significantly elevated the number of T regulatory cells in the pancreas, thus potentially controlling the autoimmune process. We show that the compounds inhibit pancreatic caspase-1 activity followed by decreased levels of the inflammatory cytokines IL-1β and IL-17 in the pancreas. These properties enable the compounds to increase the proportion of Tregs in the pancreatic lymph nodes. AS101 and SAS have been previously shown to regulate specific integrins through a unique redox mechanism. Our current results suggest that amelioration of disease in NOD mice by this unique mechanism is due to decreased infiltration of pancreatic islets combined with increased immune regulation, leading to decreased inflammation within the islets. As these tellurium compounds show remarkable lack of toxicity in clinical trials (AS101) and pre-clinical studies (SAS), they may be suitable for the treatment of type-1 diabetes.
Keywords: IL-1β; Tregs; diabetes; inflammation; integrin; tellurium.
Figures






Similar articles
-
Inhibition of α4β1 Integrin Activity by Small Tellurium Compounds Regulates PD-L1 Expression and Enhances Antitumor Effects.Int J Biol Sci. 2024 Aug 12;20(11):4407-4423. doi: 10.7150/ijbs.95350. eCollection 2024. Int J Biol Sci. 2024. PMID: 39247817 Free PMC article.
-
The effect of the novel tellurium compound AS101 on autoimmune diseases.Autoimmun Rev. 2014 Dec;13(12):1230-5. doi: 10.1016/j.autrev.2014.08.003. Epub 2014 Aug 19. Autoimmun Rev. 2014. PMID: 25153485 Review.
-
Multifunctional activity of a small tellurium redox immunomodulator compound, AS101, on dextran sodium sulfate-induced murine colitis.J Biol Chem. 2014 Jun 13;289(24):17215-27. doi: 10.1074/jbc.M113.536664. Epub 2014 Apr 24. J Biol Chem. 2014. PMID: 24764299 Free PMC article.
-
Immunomodulating tellurium compounds as anti-cancer agents.Semin Cancer Biol. 2012 Feb;22(1):60-9. doi: 10.1016/j.semcancer.2011.12.003. Epub 2011 Dec 21. Semin Cancer Biol. 2012. PMID: 22202556 Review.
-
The synthetic tellurium compound, AS101, is a novel inhibitor of IL-1beta converting enzyme.J Interferon Cytokine Res. 2007 Jun;27(6):453-62. doi: 10.1089/jir.2007.0168. J Interferon Cytokine Res. 2007. PMID: 17572009
Cited by
-
Integrins and NAFLD-associated liver diseases: clinical associations, pathophysiological mechanisms and pharmacological implications.Acta Biochim Biophys Sin (Shanghai). 2024 Sep 14;56(11):1573-1583. doi: 10.3724/abbs.2024149. Acta Biochim Biophys Sin (Shanghai). 2024. PMID: 40384047 Free PMC article. Review.
-
Gut-tropic α4β7+CD8+ T cells contribute to pancreatic β cell destruction in type 1 diabetes.Front Immunol. 2025 Jul 10;16:1623428. doi: 10.3389/fimmu.2025.1623428. eCollection 2025. Front Immunol. 2025. PMID: 40709181 Free PMC article.
-
Pancreatic draining lymph nodes (PLNs) serve as a pathogenic hub contributing to the development of type 1 diabetes.Cell Biosci. 2023 Aug 28;13(1):156. doi: 10.1186/s13578-023-01110-7. Cell Biosci. 2023. PMID: 37641145 Free PMC article. Review.
-
The tellurium-based immunomodulator, AS101 ameliorates adjuvant-induced arthritis in rats.Clin Exp Immunol. 2021 Mar;203(3):375-384. doi: 10.1111/cei.13553. Epub 2020 Dec 8. Clin Exp Immunol. 2021. PMID: 33205391 Free PMC article.
-
Inhibition of α4β1 Integrin Activity by Small Tellurium Compounds Regulates PD-L1 Expression and Enhances Antitumor Effects.Int J Biol Sci. 2024 Aug 12;20(11):4407-4423. doi: 10.7150/ijbs.95350. eCollection 2024. Int J Biol Sci. 2024. PMID: 39247817 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

