Novel Targeting to XCR1+ Dendritic Cells Using Allogeneic T Cells for Polytopical Antibody Responses in the Lymph Nodes
- PMID: 31191552
- PMCID: PMC6548820
- DOI: 10.3389/fimmu.2019.01195
Novel Targeting to XCR1+ Dendritic Cells Using Allogeneic T Cells for Polytopical Antibody Responses in the Lymph Nodes
Abstract
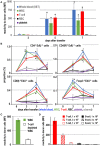
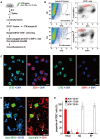
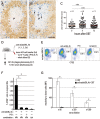
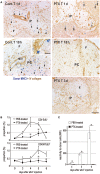
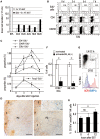
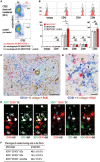
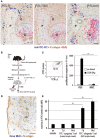
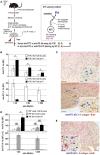
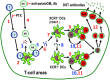
Vaccination strategy that induce efficient antibody responses polytopically in most lymph nodes (LNs) against infections has not been established yet. Because donor-specific blood transfusion induces anti-donor class I MHC antibody production in splenectomized rats, we examined the mechanism and significance of this response. Among the donor blood components, T cells were the most efficient immunogens, inducing recipient T cell and B cell proliferative responses not only in the spleen, but also in the peripheral and gut LNs. Donor T cells soon migrated to the splenic T cell area and the LNs, with a temporary significant increase in recipient NK cells. XCR1+ resident dendritic cells (DCs), but not XCR1- DCs, selectively phagocytosed donor class I MHC+ fragments after 1 day. After 1.5 days, both DC subsets formed clusters with recipient CD4+ T cells, which proliferated within these clusters. Inhibition of donor T cell migration or depletion of NK cells by pretreatment with pertussis toxin or anti-asialoGM1 antibody, respectively, significantly suppressed DC phagocytosis and subsequent immune responses. Three allogeneic strains with different NK activities had the same response but with different intensity. Donor T cell proliferation was not required, indicating that the graft vs. host reaction is dispensable. Intravenous transfer of antigen-labeled and mitotic inhibitor-treated allogeneic, but not syngeneic, T cells induced a polytopical antibody response to labeled antigens in the LNs of splenectomized rats. These results demonstrate a novel mechanism of alloresponses polytopically in the secondary lymphoid organs (SLOs) induced by allogeneic T cells. Donor T cells behave as self-migratory antigen ferries to be delivered to resident XCR1+ DCs with negligible commitment of migratory DCs. Allogeneic T cells may be clinically applicable as vaccine vectors for polytopical prophylactic antibody production even in asplenic or hyposplenic individuals.
Keywords: XCR1+ dendritic cell; allogeneic T cell; asplenia; dendritic cell targeting; lymph nodes; polytopical antibody production; vaccination.
Figures

Similar articles
-
Suppression of liver transplant rejection by anti-donor MHC antibodies via depletion of donor immunogenic dendritic cells.Int Immunol. 2021 Apr 22;33(5):261-272. doi: 10.1093/intimm/dxaa076. Int Immunol. 2021. PMID: 33258927 Free PMC article.
-
Single blood transfusion induces the production of donor-specific alloantibodies and regulatory T cells mainly in the spleen.Int Immunol. 2018 Mar 8;30(2):53-67. doi: 10.1093/intimm/dxx078. Int Immunol. 2018. PMID: 29361165 Free PMC article.
-
Systemic transmigration of allosensitizing donor dendritic cells to host secondary lymphoid organs after rat liver transplantation.Hepatology. 2008 Apr;47(4):1352-62. doi: 10.1002/hep.22161. Hepatology. 2008. PMID: 18220273
-
The microstructure of secondary lymphoid organs that support immune cell trafficking.Arch Histol Cytol. 2010;73(1):1-21. doi: 10.1679/aohc.73.1. Arch Histol Cytol. 2010. PMID: 21471663 Review.
-
Uptake of antigens from the intestine by dendritic cells.Ann N Y Acad Sci. 2004 Dec;1029:75-82. doi: 10.1196/annals.1309.010. Ann N Y Acad Sci. 2004. PMID: 15681746 Review.
Cited by
-
Engineered cellular immunotherapies in cancer and beyond.Nat Med. 2022 Apr;28(4):678-689. doi: 10.1038/s41591-022-01765-8. Epub 2022 Apr 19. Nat Med. 2022. PMID: 35440724 Free PMC article. Review.
-
Selective involution of thymic medulla by cyclosporine A with a decrease of mature thymic epithelia, XCR1+ dendritic cells, and epithelium-free areas containing Foxp3+ thymic regulatory T cells.Histochem Cell Biol. 2021 Aug;156(2):133-146. doi: 10.1007/s00418-021-01993-y. Epub 2021 May 16. Histochem Cell Biol. 2021. PMID: 33993340 Free PMC article.
-
Suppression of liver transplant rejection by anti-donor MHC antibodies via depletion of donor immunogenic dendritic cells.Int Immunol. 2021 Apr 22;33(5):261-272. doi: 10.1093/intimm/dxaa076. Int Immunol. 2021. PMID: 33258927 Free PMC article.
-
Dendritic Cell Vaccines for Cancer Immunotherapy: The Role of Human Conventional Type 1 Dendritic Cells.Pharmaceutics. 2020 Feb 15;12(2):158. doi: 10.3390/pharmaceutics12020158. Pharmaceutics. 2020. PMID: 32075343 Free PMC article. Review.
-
Stromal modifying CHST15 siRNA enhances antitumor effect synergistically with anti-PD-1 immune checkpoint antibody in murine pancreatic cancer.Sci Rep. 2025 Jul 1;15(1):20365. doi: 10.1038/s41598-025-09445-6. Sci Rep. 2025. PMID: 40595396 Free PMC article.
References
-
- Siegrist CA. Vaccine immunology. In Vaccines. 5th ed. Plotkin SA, Orenstein RW, Offit PA, Edwards KM. editors. New York, NY: Saunders Elsevier; (2008). p. 17–36.
-
- Davies JM, Lewis MP, Wimperis J, Rafi I, Ladhani S, Bolton-Maggs PH, et al. . Review of guidelines for the prevention and treatment of infection in patients with an absent or dysfunctional spleen: prepared on behalf of the British Committee for Standards in Haematology by a working party of the haemato-oncology task force. Br J Haematol. (2011) 155:308–17. 10.1111/j.1365-2141.2011.08843.x - DOI - PubMed
-
- Standring S. Gray's Anatomy, 39th ed. The Anatomical Basis of Clinical Practice. In: Blood, Lymphoid Tissues and Haemopoiesis. Churchill, MB: Livingstone; (2008). p. 75.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

