Experimental colitis in IL-10-deficient mice ameliorates in the absence of PTPN22
- PMID: 31194881
- PMCID: PMC6693971
- DOI: 10.1111/cei.13339
Experimental colitis in IL-10-deficient mice ameliorates in the absence of PTPN22
Abstract
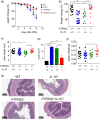
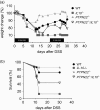
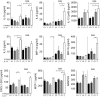
Interleukin (IL)-10 plays a key role in controlling intestinal inflammation. IL-10-deficient mice and patients with mutations in IL-10 or its receptor, IL-10R, show increased susceptibility to inflammatory bowel diseases (IBD). Protein tyrosine phosphatase, non-receptor type 22 (PTPN22) controls immune cell activation and the equilibrium between regulatory and effector T cells, playing an important role in controlling immune homoeostasis of the gut. Here, we examined the role of PTPN22 in intestinal inflammation of IL-10-deficient (IL-10-/- ) mice. We crossed IL-10-/- mice with PTPN22-/- mice to generate PTPN22-/- IL-10-/- double knock-out mice and induced colitis with dextran sodium sulphate (DSS). In line with previous reports, DSS-induced acute and chronic colitis was exacerbated in IL-10-/- mice compared to wild-type (WT) controls. However, PTPN22-/- IL-10-/- double knock-out mice developed milder disease compared to IL-10-/- mice. IL-17-promoting innate cytokines and T helper type 17 (Th17) cells were markedly increased in PTPN22-/- IL-10-/- mice, but did not provide a protctive function. CXCL1/KC was also increased in PTPN22-/- IL-10-/- mice, but therapeutic injection of CXCL1/KC in IL-10-/- mice did not ameliorate colitis. These results show that PTPN22 promotes intestinal inflammation in IL-10-deficient mice, suggesting that therapeutic targeting of PTPN22 might be beneficial in patients with IBD and mutations in IL-10 and IL-10R.
Keywords: IL-10; PTPN22; colitis; inflammatory bowel diseases (IBD).
© 2019 British Society for Immunology.
Conflict of interest statement
The authors have declared that no conflicts of interest exist.
Figures




Similar articles
-
Heme oxygenase-1 ameliorates dextran sulfate sodium-induced acute murine colitis by regulating Th17/Treg cell balance.J Biol Chem. 2014 Sep 26;289(39):26847-26858. doi: 10.1074/jbc.M114.590554. Epub 2014 Aug 11. J Biol Chem. 2014. PMID: 25112868 Free PMC article.
-
Protein tyrosine phosphatase non-receptor type 22 modulates colitis in a microbiota-dependent manner.J Clin Invest. 2019 May 20;129(6):2527-2541. doi: 10.1172/JCI123263. eCollection 2019 May 20. J Clin Invest. 2019. PMID: 31107248 Free PMC article.
-
Exacerbated intestinal inflammation in P2Y6 deficient mice is associated with Th17 activation.Biochim Biophys Acta Mol Basis Dis. 2019 Oct 1;1865(10):2595-2605. doi: 10.1016/j.bbadis.2019.06.019. Epub 2019 Jul 2. Biochim Biophys Acta Mol Basis Dis. 2019. PMID: 31271845
-
IL-23 in inflammatory bowel diseases and colon cancer.Cytokine Growth Factor Rev. 2019 Feb;45:1-8. doi: 10.1016/j.cytogfr.2018.12.002. Epub 2018 Dec 12. Cytokine Growth Factor Rev. 2019. PMID: 30563755 Review.
-
Inflamed gut mucosa: downstream of interleukin-10.Eur J Clin Invest. 2012 Jan;42(1):95-109. doi: 10.1111/j.1365-2362.2011.02552.x. Epub 2011 Jun 1. Eur J Clin Invest. 2012. PMID: 21631466 Review.
Cited by
-
Reduced Follicular Regulatory T Cells in Spleen and Pancreatic Lymph Nodes of Patients With Type 1 Diabetes.Diabetes. 2021 Dec;70(12):2892-2902. doi: 10.2337/db21-0091. Epub 2021 Oct 7. Diabetes. 2021. PMID: 34620616 Free PMC article.
-
Regulatory role of short-chain fatty acids in inflammatory bowel disease.Cell Commun Signal. 2022 May 11;20(1):64. doi: 10.1186/s12964-022-00869-5. Cell Commun Signal. 2022. PMID: 35546404 Free PMC article. Review.
-
Role of Protein Tyrosine Phosphatases in Inflammatory Bowel Disease, Celiac Disease and Diabetes: Focus on the Intestinal Mucosa.Cells. 2024 Nov 29;13(23):1981. doi: 10.3390/cells13231981. Cells. 2024. PMID: 39682729 Free PMC article. Review.
-
Real-world of Limosilactobacillus reuteri in mitigation of acute experimental colitis.J Nanobiotechnology. 2025 Jan 31;23(1):65. doi: 10.1186/s12951-025-03158-8. J Nanobiotechnology. 2025. PMID: 39891249 Free PMC article.
-
Giardia increases macrophage production of the anti-inflammatory cytokine interleukin-10 in response to lipopolysaccharide via macrophage galactose binding lectin.Immunohorizons. 2025 May 30;9(7):vlaf019. doi: 10.1093/immhor/vlaf019. Immunohorizons. 2025. PMID: 40493880 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

