Combinatorial Therapy of Zinc Metallochaperones with Mutant p53 Reactivation and Diminished Copper Binding
- PMID: 31196889
- PMCID: PMC6677634
- DOI: 10.1158/1535-7163.MCT-18-1080
Combinatorial Therapy of Zinc Metallochaperones with Mutant p53 Reactivation and Diminished Copper Binding
Abstract
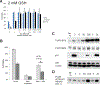
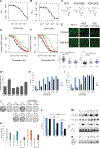
Chemotherapy and radiation are more effective in wild-type (WT) p53 tumors due to p53 activation. This is one rationale for developing drugs that reactivate mutant p53 to synergize with chemotherapy and radiation. Zinc metallochaperones (ZMC) are a new class of mutant p53 reactivators that restore WT structure and function to zinc-deficient p53 mutants. We hypothesized that the thiosemicarbazone, ZMC1, would synergize with chemotherapy and radiation. Surprisingly, this was not found. We explored the mechanism of this and found the reactive oxygen species (ROS) activity of ZMC1 negates the signal on p53 that is generated with chemotherapy and radiation. We hypothesized that a zinc scaffold generating less ROS would synergize with chemotherapy and radiation. The ROS effect of ZMC1 is generated by its chelation of redox active copper. ZMC1 copper binding (K Cu) studies reveal its affinity for copper is approximately 108 greater than Zn2+ We identified an alternative zinc scaffold (nitrilotriacetic acid) and synthesized derivatives to improve cell permeability. These compounds bind zinc in the same range as ZMC1 but bound copper much less avidly (106- to 107-fold lower) and induced less ROS. These compounds were synergistic with chemotherapy and radiation by inducing p53 signaling events on mutant p53. We explored other combinations with ZMC1 based on its mechanism of action and demonstrate that ZMC1 is synergistic with MDM2 antagonists, BCL2 antagonists, and molecules that deplete cellular reducing agents. We have identified an optimal Cu2+:Zn2+ binding ratio to facilitate development of ZMCs as chemotherapy and radiation sensitizers. Although ZMC1 is not synergistic with chemotherapy and radiation, it is synergistic with a number of other targeted agents.
©2019 American Association for Cancer Research.
Conflict of interest statement
Conflict of Interest Statement
Darren Carpizo, S. David Kimball, and Stewart Loh are affiliated with Z53 Therapeutics, Inc. as founder (DRC) and scientific board members (SDK, SNL) respectively. The other authors declare no potential conflicts of interest.
Figures



Similar articles
-
Thiosemicarbazones Functioning as Zinc Metallochaperones to Reactivate Mutant p53.Mol Pharmacol. 2017 Jun;91(6):567-575. doi: 10.1124/mol.116.107409. Epub 2017 Mar 20. Mol Pharmacol. 2017. PMID: 28320780 Free PMC article.
-
Small molecule restoration of wildtype structure and function of mutant p53 using a novel zinc-metallochaperone based mechanism.Oncotarget. 2014 Oct 15;5(19):8879-92. doi: 10.18632/oncotarget.2432. Oncotarget. 2014. PMID: 25294809 Free PMC article.
-
Zinc Metallochaperones Reactivate Mutant p53 Using an ON/OFF Switch Mechanism: A New Paradigm in Cancer Therapeutics.Clin Cancer Res. 2018 Sep 15;24(18):4505-4517. doi: 10.1158/1078-0432.CCR-18-0822. Epub 2018 Jun 18. Clin Cancer Res. 2018. PMID: 29914895 Free PMC article.
-
Reactivating mutant p53 using small molecules as zinc metallochaperones: awakening a sleeping giant in cancer.Drug Discov Today. 2015 Nov;20(11):1391-7. doi: 10.1016/j.drudis.2015.07.006. Epub 2015 Jul 20. Drug Discov Today. 2015. PMID: 26205328 Free PMC article. Review.
-
Zinc Metallochaperones as Mutant p53 Reactivators: A New Paradigm in Cancer Therapeutics.Cancers (Basel). 2018 May 29;10(6):166. doi: 10.3390/cancers10060166. Cancers (Basel). 2018. PMID: 29843463 Free PMC article. Review.
Cited by
-
Modulation of Intracellular Copper Levels as the Mechanism of Action of Anticancer Copper Complexes: Clinical Relevance.Biomedicines. 2021 Jul 21;9(8):852. doi: 10.3390/biomedicines9080852. Biomedicines. 2021. PMID: 34440056 Free PMC article. Review.
-
Understanding the prion-like behavior of mutant p53 proteins in triple-negative breast cancer pathogenesis: The current therapeutic strategies and future directions.Heliyon. 2024 Feb 10;10(4):e26260. doi: 10.1016/j.heliyon.2024.e26260. eCollection 2024 Feb 29. Heliyon. 2024. PMID: 38390040 Free PMC article. Review.
-
Biological Effects of HDAC Inhibitors Vary with Zinc Binding Group: Differential Effects on Zinc Bioavailability, ROS Production, and R175H p53 Mutant Protein Reactivation.Biomolecules. 2023 Oct 28;13(11):1588. doi: 10.3390/biom13111588. Biomolecules. 2023. PMID: 38002270 Free PMC article.
-
Alzheimer's Disease and Cancer: Common Targets.Mini Rev Med Chem. 2024;24(10):983-1000. doi: 10.2174/0113895575263108231031132404. Mini Rev Med Chem. 2024. PMID: 38037912 Review.
-
p53 and Zinc: A Malleable Relationship.Front Mol Biosci. 2022 Apr 13;9:895887. doi: 10.3389/fmolb.2022.895887. eCollection 2022. Front Mol Biosci. 2022. PMID: 35495631 Free PMC article. Review.
References
-
- Lowe SW, Bodis S, McClatchey A, Remington L, Ruley HE, Fisher DE, et al. p53 status and the efficacy of cancer therapy in vivo. Science 1994;266(5186):807–10. - PubMed
-
- Lowe SW, Ruley HE, Jacks T, Housman DE. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 1993;74(6):957–67. - PubMed
-
- Lakin ND, Jackson SP. Regulation of p53 in response to DNA damage. Oncogene 1999;18(53):7644–55. - PubMed
-
- Barbieri E, Mehta P, Chen Z, Zhang L, Slack A, Berg S, et al. MDM2 inhibition sensitizes neuroblastoma to chemotherapy-induced apoptotic cell death. Mol Cancer Ther 2006;5(9):2358–65. - PubMed
-
- Bykov VJ, Zache N, Stridh H, Westman J, Bergman J, Selivanova G, et al. PRIMA-1(MET) synergizes with cisplatin to induce tumor cell apoptosis. Oncogene 2005;24(21):3484–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

