The zinc transporter Zip14 (SLC39a14) affects Beta-cell Function: Proteomics, Gene expression, and Insulin secretion studies in INS-1E cells
- PMID: 31197210
- PMCID: PMC6565745
- DOI: 10.1038/s41598-019-44954-1
The zinc transporter Zip14 (SLC39a14) affects Beta-cell Function: Proteomics, Gene expression, and Insulin secretion studies in INS-1E cells
Abstract
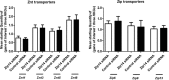
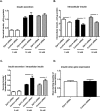
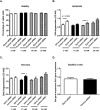
Insulin secretion from pancreatic beta-cells is dependent on zinc ions as essential components of insulin crystals, zinc transporters are thus involved in the insulin secretory process. Zip14 (SLC39a14) is a zinc importing protein that has an important role in glucose homeostasis. Zip14 knockout mice display hyperinsulinemia and impaired insulin secretion in high glucose conditions. Endocrine roles for Zip14 have been established in adipocytes and hepatocytes, but not yet confirmed in beta-cells. In this study, we investigated the role of Zip14 in the INS-1E beta-cell line. Zip14 mRNA was upregulated during high glucose stimulation and Zip14 silencing led to increased intracellular insulin content. Large-scale proteomics showed that Zip14 silencing down-regulated ribosomal mitochondrial proteins, many metal-binding proteins, and others involved in oxidative phosphorylation and insulin secretion. Furthermore, proliferation marker Mki67 was down-regulated in Zip14 siRNA-treated cells. In conclusion, Zip14 gene expression is glucose sensitive and silencing of Zip14 directly affects insulin processing in INS-1E beta-cells. A link between Zip14 and ribosomal mitochondrial proteins suggests altered mitochondrial RNA translation, which could disturb mitochondrial function and thereby insulin secretion. This highlights a role for Zip14 in beta-cell functioning and suggests Zip14 as a future pharmacological target in the treatment of beta-cell dysfunction.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Absence of Slc39a14/Zip14 in mouse pancreatic beta cells results in hyperinsulinemia.Am J Physiol Endocrinol Metab. 2024 Jan 1;326(1):E92-E105. doi: 10.1152/ajpendo.00117.2023. Epub 2023 Nov 29. Am J Physiol Endocrinol Metab. 2024. PMID: 38019082 Free PMC article.
-
SLC30A3 responds to glucose- and zinc variations in beta-cells and is critical for insulin production and in vivo glucose-metabolism during beta-cell stress.PLoS One. 2009 May 25;4(5):e5684. doi: 10.1371/journal.pone.0005684. PLoS One. 2009. PMID: 19492079 Free PMC article.
-
Effects of zinc supplementation and zinc chelation on in vitro β-cell function in INS-1E cells.BMC Res Notes. 2014 Feb 7;7:84. doi: 10.1186/1756-0500-7-84. BMC Res Notes. 2014. PMID: 24502363 Free PMC article.
-
Zinc and diabetes.Arch Biochem Biophys. 2016 Dec 1;611:79-85. doi: 10.1016/j.abb.2016.05.022. Epub 2016 Jun 1. Arch Biochem Biophys. 2016. PMID: 27262257 Review.
-
Influence of ZIP14 (slc39A14) on intestinal zinc processing and barrier function.Am J Physiol Gastrointest Liver Physiol. 2015 Feb 1;308(3):G171-8. doi: 10.1152/ajpgi.00021.2014. Epub 2014 Nov 26. Am J Physiol Gastrointest Liver Physiol. 2015. PMID: 25428902 Free PMC article. Review.
Cited by
-
Modulation of Zinc Transporter Expressions by Additional Zinc in C2C12 Cells Cultured in a High Glucose Environment and in the Presence of Insulin or Interleukin-6.Biol Trace Elem Res. 2023 Jul;201(7):3428-3437. doi: 10.1007/s12011-022-03443-9. Epub 2022 Oct 13. Biol Trace Elem Res. 2023. PMID: 36227447
-
Iron Metabolism in Pancreatic Beta-Cell Function and Dysfunction.Cells. 2021 Oct 22;10(11):2841. doi: 10.3390/cells10112841. Cells. 2021. PMID: 34831062 Free PMC article. Review.
-
Ferroptosis: a new strategy for Chinese herbal medicine treatment of diabetic nephropathy.Front Endocrinol (Lausanne). 2023 Jun 9;14:1188003. doi: 10.3389/fendo.2023.1188003. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37361521 Free PMC article. Review.
-
Role of zinc in health and disease.Clin Exp Med. 2024 Feb 17;24(1):38. doi: 10.1007/s10238-024-01302-6. Clin Exp Med. 2024. PMID: 38367035 Free PMC article. Review.
-
ZnT8 Haploinsufficiency Impacts MIN6 Cell Zinc Content and β-Cell Phenotype via ZIP-ZnT8 Coregulation.Int J Mol Sci. 2019 Nov 4;20(21):5485. doi: 10.3390/ijms20215485. Int J Mol Sci. 2019. PMID: 31690008 Free PMC article.
References
-
- Chimienti, F., Rutter, G. A., Wheeler, M. B. & Wijesekara, N. Zinc and diabetes in Zinc in Human Health (ed. Rink, L.) 493–513 (IOS Press, 2011).
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

