New injectable two-step forming hydrogel for delivery of bioactive substances in tissue regeneration
- PMID: 31198583
- PMCID: PMC6547312
- DOI: 10.1093/rb/rbz018
New injectable two-step forming hydrogel for delivery of bioactive substances in tissue regeneration
Abstract
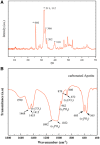
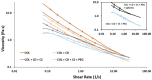
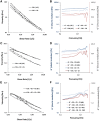
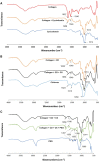
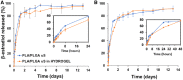
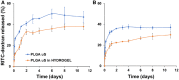
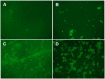
A hydrogel based on chitosan, collagen, hydroxypropyl-γ-cyclodextrin and polyethylene glycol was developed and characterized. The incorporation of nano-hydroxyapatite and pre-encapsulated hydrophobic/hydrophilic model drugs diminished the porosity of hydrogel from 81.62 ± 2.25% to 69.98 ± 3.07%. Interactions between components of hydrogel, demonstrated by FTIR spectroscopy and rheology, generated a network that was able to trap bioactive components and delay the burst delivery. The thixotropic behavior of hydrogel provided adaptability to facilitate its implantation in a minimally invasive way. Release profiles from microspheres included or not in hydrogel revealed a two-phase behavior with a burst- and a controlled-release period. The same release rate for microspheres included or not in the hydrogel in the controlled-release period demonstrated that mass transfer process was controlled by internal diffusion. Effective diffusion coefficients, D eff, that describe internal diffusion inside microspheres, and mass transfer coefficients, h, i.e. the contribution of hydrogel to mass transfer, were determined using 'genetic algorithms', obtaining values between 2.64·10-15 and 6.67·10-15 m2/s for D eff and 8.50·10-10 to 3.04·10-9 m/s for h. The proposed model fits experimental data, obtaining an R 2-value ranged between 95.41 and 98.87%. In vitro culture of mesenchymal stem cells in hydrogel showed no manifestations of intolerance or toxicity, observing an intense proliferation of the cells after 7 days, being most of the scaffold surface occupied by living cells.
Keywords: FITC-dextran; collagen–cyclodextrin–chitosan; estradiol; hydrogel; mass transfer; rheology.
Figures



Similar articles
-
Pharmacologically active microcarriers delivering BDNF within a hydrogel: Novel strategy for human bone marrow-derived stem cells neural/neuronal differentiation guidance and therapeutic secretome enhancement.Acta Biomater. 2017 Feb;49:167-180. doi: 10.1016/j.actbio.2016.11.030. Epub 2016 Nov 16. Acta Biomater. 2017. PMID: 27865962
-
Nanostructured gellan and xanthan hydrogel depot integrated within a baghdadite scaffold augments bone regeneration.J Tissue Eng Regen Med. 2017 Apr;11(4):1195-1211. doi: 10.1002/term.2023. Epub 2015 Apr 7. J Tissue Eng Regen Med. 2017. PMID: 25846217
-
Bioactive injectable triple acting thermosensitive hydrogel enriched with nano-hydroxyapatite for bone regeneration: in-vitro characterization, Saos-2 cell line cell viability and osteogenic markers evaluation.Drug Dev Ind Pharm. 2019 May;45(5):787-804. doi: 10.1080/03639045.2019.1572184. Epub 2019 Feb 4. Drug Dev Ind Pharm. 2019. Retraction in: Drug Dev Ind Pharm. 2022 Dec;48(12):746. doi: 10.1080/03639045.2023.2165803. PMID: 30672348 Retracted.
-
Preparation and characterization of novel semi-interpenetrating polymer network hydrogel microspheres of chitosan and hydroxypropyl cellulose for controlled release of chlorothiazide.J Microencapsul. 2009 Feb;26(1):27-36. doi: 10.1080/02652040802109770. J Microencapsul. 2009. PMID: 18608812
-
Repair of osteochondral defects using injectable chitosan-based hydrogel encapsulated synovial fluid-derived mesenchymal stem cells in a rabbit model.Mater Sci Eng C Mater Biol Appl. 2019 Jun;99:541-551. doi: 10.1016/j.msec.2019.01.115. Epub 2019 Jan 26. Mater Sci Eng C Mater Biol Appl. 2019. PMID: 30889728
Cited by
-
Porous PLLA microspheres dispersed in HA/collagen hydrogel as injectable facial fillers to enhance aesthetic effects.Regen Biomater. 2025 May 23;12:rbaf049. doi: 10.1093/rb/rbaf049. eCollection 2025. Regen Biomater. 2025. PMID: 40556787 Free PMC article.
-
Injectable Composite Systems Based on Microparticles in Hydrogels for Bioactive Cargo Controlled Delivery.Gels. 2021 Sep 18;7(3):147. doi: 10.3390/gels7030147. Gels. 2021. PMID: 34563033 Free PMC article. Review.
-
Mitochondria-targeting polydopamine-coated nanodrugs for effective photothermal- and chemo-synergistic therapies against lung cancer.Regen Biomater. 2022 Aug 1;9:rbac051. doi: 10.1093/rb/rbac051. eCollection 2022. Regen Biomater. 2022. PMID: 35958515 Free PMC article.
-
PLGA-BMP-2 and PLA-17β-Estradiol Microspheres Reinforcing a Composite Hydrogel for Bone Regeneration in Osteoporosis.Pharmaceutics. 2019 Dec 3;11(12):648. doi: 10.3390/pharmaceutics11120648. Pharmaceutics. 2019. PMID: 31817033 Free PMC article.
-
Advancements and Applications of Injectable Hydrogel Composites in Biomedical Research and Therapy.Gels. 2023 Jun 30;9(7):533. doi: 10.3390/gels9070533. Gels. 2023. PMID: 37504412 Free PMC article. Review.
References
-
- Peppas NA, Bures P, Leobandung W. et al. Hydrogels in pharmaceutical formulations. Eur J Pharm Biopharm 2000;50:27–46. - PubMed
-
- Hoffman AS. Hydrogels for biomedical applications. Adv Drug Deliv Rev 2002;54:3–12. - PubMed
-
- Vermonden T, Censi R, Hennink WE.. Hydrogels for protein delivery. Chem Rev 2012;112:2853–88. - PubMed
-
- Peppas NA, Hilt JZ, Khademhosseini A. et al. Hydrogels in biology and medicine: from molecular principles to bionanotechnology. Adv Mater 2006;18:1345–60.
-
- Li Y, Rodrigues J, Tomás H.. Injectable and biodegradable hydrogels: gelation, biodegradation and biomedical applications. Chem Soc Rev 2012;41:2193–221. - PubMed
LinkOut - more resources
Full Text Sources

