Effect of solution condition on hydroxyapatite formation in evaluating bioactivity of B2O3 containing 45S5 bioactive glasses
- PMID: 31198889
- PMCID: PMC6555879
- DOI: 10.1016/j.bioactmat.2019.05.002
Effect of solution condition on hydroxyapatite formation in evaluating bioactivity of B2O3 containing 45S5 bioactive glasses
Abstract
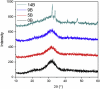
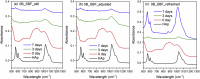
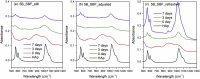
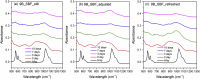
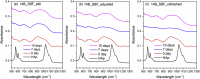
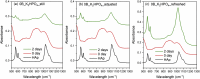
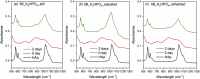
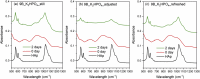
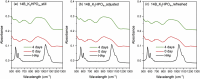
The effects of testing solutions and conditions on hydroxyapatite (HAp) formation as a means of in vitro bioactivity evaluation of B2O3 containing 45S5 bioactive glasses were systematically investigated. Four glass samples prepared by the traditional melt and quench process, where SiO2 in 45S5 was gradually replaced by B2O3 (up to 30%), were studied. Two solutions: the simulated body fluid (SBF) and K2HPO4 solutions were used as the medium for evaluating in vitro bioactivity through the formation of HAp on glass surface as a function of time. It was found that addition of boron oxide delayed the HAp formation in both SBF and K2HPO4 solutions, while the reaction between glass and the K2HPO4 solution is much faster as compared to SBF. In addition to the composition and medium effects, we also studied whether the solution treatments (e.g., adjusting to maintain a pH of 7.4, refreshing solution at certain time interval, and no disturbance during immersion) affect HAp formation. Fourier transform infrared spectrometer (FTIR) equipped with an attenuated total reflection (ATR) sampling technique and scanning electron microscopy (SEM) were conducted to identify HAp formation on glass powder surfaces and to observe HAp morphologies, respectively. The results show that refreshing solution every 24 h produced the fastest HAp formation for low boron-containing samples when SBF was used as testing solution, while no significant differences were observed when K2HPO4 solution was used. This study thus suggests the testing solutions and conditions play an important role on the in vitro bioactivity testing results and should be carefully considered when study materials with varying bioactivities.
Keywords: Bioactive glasses; Boron oxide; Hydroxyapatite; In vitro.
Figures



Similar articles
-
Mixed Network Former Effect on Structure, Physical Properties, and Bioactivity of 45S5 Bioactive Glasses: An Integrated Experimental and Molecular Dynamics Simulation Study.J Phys Chem B. 2018 Mar 8;122(9):2564-2577. doi: 10.1021/acs.jpcb.7b12127. Epub 2018 Feb 23. J Phys Chem B. 2018. PMID: 29390190
-
In vitro bioactivity evaluation, mechanical properties and microstructural characterization of Na₂O-CaO-B₂O₃-P₂O₅ glasses.Spectrochim Acta A Mol Biomol Spectrosc. 2015 Jun 5;144:88-98. doi: 10.1016/j.saa.2015.02.076. Epub 2015 Feb 26. Spectrochim Acta A Mol Biomol Spectrosc. 2015. PMID: 25748986
-
Assessment of in vitro bioactivity of SiO2-BaO-ZnO-B2O3-Al2O3 glasses: An optico-analytical approach.Mater Sci Eng C Mater Biol Appl. 2012 Oct 1;32(7):1941-1947. doi: 10.1016/j.msec.2012.05.034. Epub 2012 May 27. Mater Sci Eng C Mater Biol Appl. 2012. PMID: 34062679
-
Gallium-containing phospho-silicate glasses: synthesis and in vitro bioactivity.Mater Sci Eng C Mater Biol Appl. 2012 Aug 1;32(6):1401-6. doi: 10.1016/j.msec.2012.04.016. Epub 2012 Apr 20. Mater Sci Eng C Mater Biol Appl. 2012. PMID: 24364938
-
Composite bone cements loaded with a bioactive and ferrimagnetic glass-ceramic: Leaching, bioactivity and cytocompatibility.Mater Sci Eng C Mater Biol Appl. 2015 Aug;53:95-103. doi: 10.1016/j.msec.2015.03.039. Epub 2015 Mar 24. Mater Sci Eng C Mater Biol Appl. 2015. PMID: 26042695 Review.
Cited by
-
Multifunctional zinc ion doped sol - gel derived mesoporous bioactive glass nanoparticles for biomedical applications.Bioact Mater. 2019 Oct 27;4:312-321. doi: 10.1016/j.bioactmat.2019.10.002. eCollection 2019 Dec. Bioact Mater. 2019. PMID: 31709314 Free PMC article.
-
Zinc-Doped Bioactive Glass/Polycaprolactone Hybrid Scaffolds Manufactured by Direct and Indirect 3D Printing Methods for Bone Regeneration.Cells. 2023 Jun 30;12(13):1759. doi: 10.3390/cells12131759. Cells. 2023. PMID: 37443794 Free PMC article.
-
Ginger extract loaded Fe2O3/MgO-doped hydroxyapatite: Evaluation of biological properties for bone-tissue engineering.J Am Ceram Soc. 2024 Apr;107(4):2081-2092. doi: 10.1111/jace.19568. Epub 2023 Dec 8. J Am Ceram Soc. 2024. PMID: 38855017 Free PMC article.
-
Comparative In Vitro Dissolution Assessment of Calcined and Uncalcined Hydroxyapatite Using Differences in Bioresorbability and Biomineralization.Int J Mol Sci. 2024 Jan 3;25(1):621. doi: 10.3390/ijms25010621. Int J Mol Sci. 2024. PMID: 38203791 Free PMC article.
-
Mesh Ti6Al4V Material Manufactured by Selective Laser Melting (SLM) as a Promising Intervertebral Fusion Cage.Int J Mol Sci. 2022 Apr 3;23(7):3985. doi: 10.3390/ijms23073985. Int J Mol Sci. 2022. PMID: 35409345 Free PMC article.
References
-
- Hench L.L., Splinter R.J., Allen W.C., Greenlee T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971;5:117–141.
-
- Hench L.L. The story of Bioglass. J. Mater. Sci. Mater. Med. 2006;17:967–978. - PubMed
-
- Hench L.L. Chronology of bioactive glass development and clinical applications. New J. Glass Ceram. 2013;03:67–73.
-
- Jones J.R. Reprint of: review of bioactive glass: from Hench to hybrids. Acta Biomater. 2015;23:S53–S82. - PubMed
-
- Rabiee S.M., Nazparvar N., Azizian M., Vashaee D., Tayebi L. Effect of ion substitution on properties of bioactive glasses: a review. Ceram. Int. 2015;41:7241–7251.
LinkOut - more resources
Full Text Sources
Miscellaneous

