O-GlcNAcylation in immunity and inflammation: An intricate system (Review)
- PMID: 31198979
- PMCID: PMC6605495
- DOI: 10.3892/ijmm.2019.4238
O-GlcNAcylation in immunity and inflammation: An intricate system (Review)
Abstract
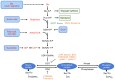
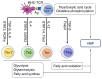
Chronic, low‑grade inflammation associated with obesity and diabetes result from the infiltration of adipose and vascular tissue by immune cells and contributes to cardiovascular complications. Despite an incomplete understanding of the mechanistic underpinnings of immune cell differentiation and inflammation, O‑GlcNAcylation, the addition of O‑linked N‑acetylglucosamine (O‑GlcNAc) to cytoplasmic, nuclear and mitochondrial proteins by the two cycling enzymes, the O‑linked N‑acetylglucosamine transferase (OGT) and the O‑GlcNAcase (OGA), may contribute to fine‑tune immunity and inflammation in both physiological and pathological conditions. Early studies have indicated that O‑GlcNAcylation of proteins play a pro‑inflammatory role in diabetes and insulin resistance, whereas subsequent studies have demonstrated that this post‑translational modification could also be protective against acute injuries. These studies suggest that diverse types of insults result in dynamic changes to O‑GlcNAcylation patterns, which fluctuate with cellular metabolism to promote or inhibit inflammation. In this review, the current understanding of O‑GlcNAcylation and its adaptive modulation in immune and inflammatory responses is summarized.
Figures
Similar articles
-
Cisplatin enhances protein O‑GlcNAcylation by altering the activity of OGT, OGA and AMPK in human non‑small cell lung cancer cells.Int J Oncol. 2021 Jun;58(6):27. doi: 10.3892/ijo.2021.5207. Epub 2021 Apr 13. Int J Oncol. 2021. PMID: 33846785
-
O‑GlcNAcylation contributes to intermittent hypoxia‑associated vascular dysfunction via modulation of MAPKs but not CaMKII pathways.Mol Med Rep. 2021 Nov;24(5):744. doi: 10.3892/mmr.2021.12384. Epub 2021 Aug 26. Mol Med Rep. 2021. PMID: 34435655 Free PMC article.
-
Too sweet to resist: Control of immune cell function by O-GlcNAcylation.Cell Immunol. 2018 Nov;333:85-92. doi: 10.1016/j.cellimm.2018.05.010. Epub 2018 Jun 2. Cell Immunol. 2018. PMID: 29887419 Free PMC article. Review.
-
The Role of O-GlcNAcylation in Immune Cell Activation.Front Endocrinol (Lausanne). 2021 Apr 27;12:596617. doi: 10.3389/fendo.2021.596617. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33986724 Free PMC article. Review.
-
O-GlcNAcylation promotes migration and invasion in human ovarian cancer cells via the RhoA/ROCK/MLC pathway.Mol Med Rep. 2017 Apr;15(4):2083-2089. doi: 10.3892/mmr.2017.6244. Epub 2017 Feb 24. Mol Med Rep. 2017. PMID: 28259907 Free PMC article.
Cited by
-
Mechanistic roles for altered O-GlcNAcylation in neurodegenerative disorders.Biochem J. 2021 Jul 30;478(14):2733-2758. doi: 10.1042/BCJ20200609. Biochem J. 2021. PMID: 34297044 Free PMC article. Review.
-
Targeted Protein O-GlcNAcylation Using Bifunctional Small Molecules.J Am Chem Soc. 2024 Apr 10;146(14):9779-9789. doi: 10.1021/jacs.3c14380. Epub 2024 Apr 1. J Am Chem Soc. 2024. PMID: 38561350 Free PMC article.
-
Downregulation of O-GlcNAcylation enhances etoposide-induced p53-mediated apoptosis in HepG2 human liver cancer cells.FEBS Open Bio. 2025 Jul;15(7):1176-1188. doi: 10.1002/2211-5463.70028. Epub 2025 Apr 16. FEBS Open Bio. 2025. PMID: 40237201 Free PMC article.
-
O-GlcNAcylation: The Underestimated Emerging Regulators of Skeletal Muscle Physiology.Cells. 2022 May 30;11(11):1789. doi: 10.3390/cells11111789. Cells. 2022. PMID: 35681484 Free PMC article. Review.
-
Nutrient Sensor mTOR and OGT: Orchestrators of Organelle Homeostasis in Pancreatic β-Cells.J Diabetes Res. 2020 Dec 16;2020:8872639. doi: 10.1155/2020/8872639. eCollection 2020. J Diabetes Res. 2020. PMID: 33457426 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

