Aims, Study Design, and Enrollment Results From the Assessing Predictors of Infant Respiratory Syncytial Virus Effects and Severity Study
- PMID: 31199303
- PMCID: PMC6595944
- DOI: 10.2196/12907
Aims, Study Design, and Enrollment Results From the Assessing Predictors of Infant Respiratory Syncytial Virus Effects and Severity Study
Abstract
Background: The majority of infants hospitalized with primary respiratory syncytial virus (RSV) infection have no obvious risk factors for severe disease.
Objective: The aim of this study (Assessing Predictors of Infant RSV Effects and Severity, AsPIRES) was to identify factors associated with severe disease in full-term healthy infants younger than 10 months with primary RSV infection.
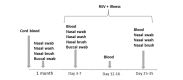
Methods: RSV infected infants were enrolled from 3 cohorts during consecutive winters from August 2012 to April 2016 in Rochester, New York. A birth cohort was prospectively enrolled and followed through their first winter for development of RSV infection. An outpatient supplemental cohort was enrolled in the emergency department or pediatric offices, and a hospital cohort was enrolled on admission with RSV infection. RSV was diagnosed by reverse transcriptase-polymerase chain reaction. Demographic and clinical data were recorded and samples collected for assays: buccal swab (cytomegalovirus polymerase chain reaction, PCR), nasal swab (RSV qualitative PCR, complete viral gene sequence, 16S ribosomal ribonucleic acid [RNA] amplicon microbiota analysis), nasal wash (chemokine and cytokine assays), nasal brush (nasal respiratory epithelial cell gene expression using RNA sequencing [RNAseq]), and 2 to 3 ml of heparinized blood (flow cytometry, RNAseq analysis of purified cluster of differentiation [CD]4+, CD8+, B cells and natural killer cells, and RSV-specific antibody). Cord blood (RSV-specific antibody) was also collected for the birth cohort. Univariate and multivariate logistic regression will be used for analysis of data using a continuous Global Respiratory Severity Score (GRSS) as the outcome variable. Novel statistical methods will be developed for integration of the large complex datasets.
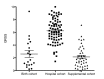
Results: A total of 453 infants were enrolled into the 3 cohorts; 226 in the birth cohort, 60 in the supplemental cohort, and 78 in the hospital cohort. A total of 126 birth cohort infants remained in the study and were evaluated for 150 respiratory illnesses. Of the 60 RSV positive infants in the supplemental cohort, 42 completed the study, whereas all 78 of the RSV positive hospital cohort infants completed the study. A GRSS was calculated for each RSV-infected infant and is being used to analyze each of the complex datasets by correlation with disease severity in univariate and multivariate methods.
Conclusions: The AsPIRES study will provide insights into the complex pathogenesis of RSV infection in healthy full-term infants with primary RSV infection. The analysis will allow assessment of multiple factors potentially influencing the severity of RSV infection including the level of RSV specific antibodies, the innate immune response of nasal epithelial cells, the adaptive response by various lymphocyte subsets, the resident airway microbiota, and viral factors. Results of this study will inform disease interventions such as vaccines and antiviral therapies.
Keywords: T-lymphocytes; gene expression; immunoglobulins; innate immunity; microbiota; respiratory syncytial virus; transcriptome.
©Edward E Walsh, Thomas J Mariani, ChinYi Chu, Alex Grier, Steven R Gill, Xing Qiu, Lu Wang, Jeanne Holden-Wiltse, Anthony Corbett, Juilee Thakar, Lauren Benoodt, Matthew N. McCall, David J Topham, Ann R Falsey, Mary T Caserta. Originally published in JMIR Research Protocols (http://www.researchprotocols.org), 06.06.2019.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures
Similar articles
-
Viral load in hospitalized infants with respiratory syncytial virus bronchiolitis: a three-way comparative analysis.Eur J Pediatr. 2024 Aug;183(8):3471-3478. doi: 10.1007/s00431-024-05614-3. Epub 2024 May 23. Eur J Pediatr. 2024. PMID: 38780651
-
T helper 1/T helper 2 cytokine imbalance in respiratory syncytial virus infection is associated with increased endogenous plasma cortisol.Pediatrics. 2006 May;117(5):e878-86. doi: 10.1542/peds.2005-2119. Epub 2006 Apr 17. Pediatrics. 2006. PMID: 16618789
-
Whole blood gene expression profiles to assess pathogenesis and disease severity in infants with respiratory syncytial virus infection.PLoS Med. 2013 Nov;10(11):e1001549. doi: 10.1371/journal.pmed.1001549. Epub 2013 Nov 12. PLoS Med. 2013. PMID: 24265599 Free PMC article.
-
Influenza and respiratory syncytial virus in infants study (IRIS) of hospitalized and non-ill infants aged <1 year in four countries: study design and methods.BMC Infect Dis. 2017 Mar 22;17(1):222. doi: 10.1186/s12879-017-2299-7. BMC Infect Dis. 2017. PMID: 28330443 Free PMC article.
-
Global disease burden of and risk factors for acute lower respiratory infections caused by respiratory syncytial virus in preterm infants and young children in 2019: a systematic review and meta-analysis of aggregated and individual participant data.Lancet. 2024 Mar 30;403(10433):1241-1253. doi: 10.1016/S0140-6736(24)00138-7. Epub 2024 Feb 14. Lancet. 2024. PMID: 38367641
Cited by
-
Unbiased analysis of peripheral blood mononuclear cells reveals CD4 T cell response to RSV matrix protein.Vaccine X. 2020 Apr 21;5:100065. doi: 10.1016/j.jvacx.2020.100065. eCollection 2020 Aug 7. Vaccine X. 2020. PMID: 32529184 Free PMC article.
-
A systems genomics approach uncovers molecular associates of RSV severity.PLoS Comput Biol. 2021 Dec 28;17(12):e1009617. doi: 10.1371/journal.pcbi.1009617. eCollection 2021 Dec. PLoS Comput Biol. 2021. PMID: 34962914 Free PMC article.
-
Temporal Dysbiosis of Infant Nasal Microbiota Relative to Respiratory Syncytial Virus Infection.J Infect Dis. 2021 May 20;223(9):1650-1658. doi: 10.1093/infdis/jiaa577. J Infect Dis. 2021. PMID: 32926147 Free PMC article.
-
Airway gene-expression classifiers for respiratory syncytial virus (RSV) disease severity in infants.BMC Med Genomics. 2021 Feb 25;14(1):57. doi: 10.1186/s12920-021-00913-2. BMC Med Genomics. 2021. PMID: 33632195 Free PMC article.
-
Airway Gene Expression Correlates of Respiratory Syncytial Virus Disease Severity and Microbiome Composition in Infants.J Infect Dis. 2021 May 20;223(9):1639-1649. doi: 10.1093/infdis/jiaa576. J Infect Dis. 2021. PMID: 32926149 Free PMC article.
References
-
- Hall CB, Weinberg GA, Iwane MK, Blumkin AK, Edwards KM, Staat MA, Auinger P, Griffin MR, Poehling KA, Erdman D, Grijalva CG, Zhu Y, Szilagyi P. The burden of respiratory syncytial virus infection in young children. N Engl J Med. 2009 Feb 5;360(6):588–98. doi: 10.1056/NEJMoa0804877. http://europepmc.org/abstract/MED/19196675 360/6/588 - DOI - PMC - PubMed
-
- Bloom-Feshbach K, Alonso WJ, Charu V, Tamerius J, Simonsen L, Miller MA, Viboud C. Latitudinal variations in seasonal activity of influenza and respiratory syncytial virus (RSV): a global comparative review. PLoS One. 2013;8(2):e54445. doi: 10.1371/journal.pone.0054445. http://dx.plos.org/10.1371/journal.pone.0054445 PONE-D-12-22234 - DOI - DOI - PMC - PubMed
-
- Haynes AK, Manangan AP, Iwane MK, Sturm-Ramirez K, Homaira N, Brooks WA, Luby S, Rahman M, Klena JD, Zhang Y, Yu H, Zhan F, Dueger E, Mansour AM, Azazzy N, McCracken JP, Bryan JP, Lopez MR, Burton DC, Bigogo G, Breiman RF, Feikin DR, Njenga K, Montgomery J, Cohen AL, Moyes J, Pretorius M, Cohen C, Venter M, Chittaganpitch M, Thamthitiwat S, Sawatwong P, Baggett HC, Luber G, Gerber SI. Respiratory syncytial virus circulation in seven countries with Global Disease Detection Regional Centers. J Infect Dis. 2013 Dec 15;208(Suppl 3):S246–54. doi: 10.1093/infdis/jit515.jit515 - DOI - PubMed
-
- Midgley CM, Haynes AK, Baumgardner JL, Chommanard C, Demas SW, Prill MM, Abedi GR, Curns AT, Watson JT, Gerber SI. Determining the seasonality of respiratory syncytial virus in the United States: the impact of increased molecular testing. J Infect Dis. 2017 Dec 1;216(3):345–55. doi: 10.1093/infdis/jix275. http://europepmc.org/abstract/MED/28859428 3860464 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

