A Pragmatic Cluster Randomized Trial of an Electronic Clinical Decision Support System to Improve Chronic Kidney Disease Management in Primary Care: Design, Rationale, and Implementation Experience
- PMID: 31199334
- PMCID: PMC6594214
- DOI: 10.2196/14022
A Pragmatic Cluster Randomized Trial of an Electronic Clinical Decision Support System to Improve Chronic Kidney Disease Management in Primary Care: Design, Rationale, and Implementation Experience
Abstract
Background: The diagnosis of chronic kidney disease (CKD) is based on laboratory results easily extracted from electronic health records; therefore, CKD identification and management is an ideal area for targeted electronic decision support efforts. Early CKD management frequently occurs in primary care settings where primary care providers (PCPs) may not implement all the best practices to prevent CKD-related complications. Few previous studies have employed randomized trials to assess a CKD electronic clinical decision support system (eCDSS) that provided recommendations to PCPs tailored to each patient based on laboratory results.
Objective: The aim of this study was to report the trial design and implementation experience of a CKD eCDSS in primary care.
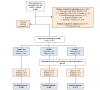
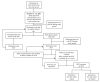
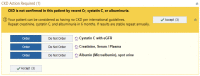
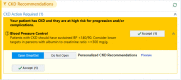
Methods: This was a 3-arm pragmatic cluster-randomized trial at an academic general internal medicine practice. Eligible patients had 2 previous estimated-glomerular-filtration-rates by serum creatinine (eGFRCr) <60 mL/min/1.73m2 at least 90 days apart. Randomization occurred at the PCP level. For patients of PCPs in either of the 2 intervention arms, the research team ordered triple-marker testing (serum creatinine, serum cystatin-c, and urine albumin-creatinine-ratio) at the beginning of the study period, to be completed when acquiring labs for regular clinical care. The eCDSS launched for PCPs and patients in the intervention arms during a regular PCP visit subsequent to completing the triple-marker testing. The eCDSS delivered individualized guidance on cardiovascular risk-reduction, potassium and proteinuria management, and patient education. Patients in the eCDSS+ arm also received a pharmacist phone call to reinforce CKD-related education. The primary clinical outcome is blood pressure change from baseline at 6 months after the end of the trial, and the main secondary outcome is provider awareness of CKD diagnosis. We also collected process, patient-centered, and implementation outcomes.
Results: A multidisciplinary team (primary care internist, nephrologists, pharmacist, and informaticist) designed the eCDSS to integrate into the current clinical workflow. All 81 PCPs contacted agreed to participate and were randomized. Of 995 patients initially eligible by eGFRCr, 413 were excluded per protocol and 58 opted out or withdrew, resulting in 524 patient participants (188 usual care; 165 eCDSS; and 171 eCDSS+). During the 12-month intervention period, 53.0% (178/336) of intervention patient participants completed triple-marker labs. Among these, 138/178 (77.5%) had a PCP appointment after the triple-marker labs resulted; the eCDSS was opened for 73.9% (102/138), with orders or education signed for 81.4% (83/102).
Conclusions: Successful integration of an eCDSS into primary care workflows and high eCDSS utilization rates at eligible visits suggest this tailored electronic approach is feasible and has the potential to improve guideline-concordant CKD care.
Trial registration: ClinicalTrials.gov NCT02925962; https://clinicaltrials.gov/ct2/show/NCT02925962 (Archived by WebCite at http://www.webcitation.org/78qpx1mjR).
International registered report identifier (irrid): DERR1-10.2196/14022.
Keywords: chronic kidney disease; clinical decision support systems; electronic health records; pragmatic clinical trial.
©Elaine C Khoong, Leah Karliner, Lowell Lo, Marilyn Stebbins, Andrew Robinson, Sarita Pathak, Jasmine Santoyo-Olsson, Rebecca Scherzer, Carmen A Peralta. Originally published in JMIR Research Protocols (http://www.researchprotocols.org), 07.06.2019.
Conflict of interest statement
Conflicts of Interest: CP is currently the chief medical officer of Cricket Health. The other authors declare they have no conflicts of interest.
Figures
Similar articles
-
Electronic Decision Support for Management of CKD in Primary Care: A Pragmatic Randomized Trial.Am J Kidney Dis. 2020 Nov;76(5):636-644. doi: 10.1053/j.ajkd.2020.05.013. Epub 2020 Jul 22. Am J Kidney Dis. 2020. PMID: 32682696 Free PMC article. Clinical Trial.
-
Implementation of a pragmatic randomized trial of screening for chronic kidney disease to improve care among non-diabetic hypertensive veterans.BMC Nephrol. 2017 Apr 12;18(1):132. doi: 10.1186/s12882-017-0541-6. BMC Nephrol. 2017. PMID: 28399844 Free PMC article. Clinical Trial.
-
Implementation of an Electronic Clinical Decision Support System for the Early Recognition and Management of Dysglycemia in an Inpatient Mental Health Setting Using CogStack: Protocol for a Pilot Hybrid Type 3 Effectiveness-Implementation Randomized Controlled Cluster Trial.JMIR Res Protoc. 2024 Apr 5;13:e49548. doi: 10.2196/49548. JMIR Res Protoc. 2024. PMID: 38578666 Free PMC article.
-
Comparing Two Approaches to Help Patients and Doctors Talk about Chronic Pain Treatments -- Voices in Pain Care [Internet].Washington (DC): Patient-Centered Outcomes Research Institute (PCORI); 2022 Apr. Washington (DC): Patient-Centered Outcomes Research Institute (PCORI); 2022 Apr. PMID: 39661719 Free Books & Documents. Review.
-
Early Identification and Management of Chronic Kidney Disease: A Narrative Review of the Crucial Role of Primary Care Practitioners.Adv Ther. 2024 Oct;41(10):3757-3770. doi: 10.1007/s12325-024-02957-z. Epub 2024 Aug 20. Adv Ther. 2024. PMID: 39162984 Free PMC article. Review.
Cited by
-
Electronic Decision Support for Management of CKD in Primary Care: A Pragmatic Randomized Trial.Am J Kidney Dis. 2020 Nov;76(5):636-644. doi: 10.1053/j.ajkd.2020.05.013. Epub 2020 Jul 22. Am J Kidney Dis. 2020. PMID: 32682696 Free PMC article. Clinical Trial.
-
Expectation of clinical decision support systems: a survey study among nephrologist end-users.BMC Med Inform Decis Mak. 2023 Oct 26;23(1):239. doi: 10.1186/s12911-023-02317-x. BMC Med Inform Decis Mak. 2023. PMID: 37884906 Free PMC article.
-
Optimal Early Diagnosis and Monitoring of Diabetic Kidney Disease in Type 2 Diabetes Mellitus: Addressing the Barriers to Albuminuria Testing.J Prim Care Community Health. 2021 Jan-Dec;12:21501327211003683. doi: 10.1177/21501327211003683. J Prim Care Community Health. 2021. PMID: 33749371 Free PMC article. Review.
-
Assessing trends and variability in outpatient dual testing for chronic kidney disease with urine albumin and serum creatinine, 2009-2018: a retrospective cohort study in the Veterans Health Administration System.BMJ Open. 2024 Feb 12;14(2):e073136. doi: 10.1136/bmjopen-2023-073136. BMJ Open. 2024. PMID: 38346884 Free PMC article.
-
A Virtual Multidisciplinary Care Program for Management of Advanced Chronic Kidney Disease: Matched Cohort Study.J Med Internet Res. 2020 Feb 12;22(2):e17194. doi: 10.2196/17194. J Med Internet Res. 2020. PMID: 32049061 Free PMC article.
References
-
- Centers for Disease Control and Prevention. [2019-05-29]. Chronic Kidney Disease Initiative https://www.cdc.gov/ckd/
-
- United States Renal Data System . American Journal of Kidney Diseases. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2018. [2019-05-31]. Morbidity and Mortality in Patients with CKD https://www.ajkd.org/article/S0272-6386(17)30077-X/pdf .
-
- Abdel-Kader K, Greer RC, Boulware LE, Unruh ML. Primary care physicians' familiarity, beliefs, and perceived barriers to practice guidelines in non-diabetic CKD: a survey study. BMC Nephrol. 2014 Apr 22;15:64. doi: 10.1186/1471-2369-15-64. https://www.biomedcentral.com/1471-2369/15/64 1471-2369-15-64 - DOI - PMC - PubMed
-
- Tuot DS, Plantinga LC, Hsu C, Jordan R, Burrows NR, Hedgeman E, Yee J, Saran R, Powe NR, Centers for Disease Control Chronic Kidney Disease Surveillance Team Chronic kidney disease awareness among individuals with clinical markers of kidney dysfunction. Clin J Am Soc Nephrol. 2011 Aug;6(8):1838–44. doi: 10.2215/CJN.00730111. http://cjasn.asnjournals.org/cgi/pmidlookup?view=long&pmid=21784832 CJN.00730111 - DOI - PMC - PubMed
-
- Peralta CA, Shlipak MG, Judd S, Cushman M, McClellan W, Zakai NA, Safford MM, Zhang X, Muntner P, Warnock D. Detection of chronic kidney disease with creatinine, cystatin C, and urine albumin-to-creatinine ratio and association with progression to end-stage renal disease and mortality. J Am Med Assoc. 2011 Apr 20;305(15):1545–52. doi: 10.1001/jama.2011.468. http://europepmc.org/abstract/MED/21482744 jama.2011.468 - DOI - PMC - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

