Advanced age promotes colonic dysfunction and gut-derived lung infection after stroke
- PMID: 31199577
- PMCID: PMC6718525
- DOI: 10.1111/acel.12980
Advanced age promotes colonic dysfunction and gut-derived lung infection after stroke
Abstract
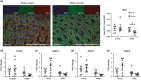
Bacterial infection a leading cause of death among patients with stroke, with elderly patients often presenting with more debilitating outcomes. The findings from our retrospective study, supported by previous clinical reports, showed that increasing age is an early predictor for developing fatal infectious complications after stroke. However, exactly how and why older individuals are more susceptible to infection after stroke remains unclear. Using a mouse model of transient ischaemic stroke, we demonstrate that older mice (>12 months) present with greater spontaneous bacterial lung infections compared to their younger counterparts (7-10 weeks) after stroke. Importantly, we provide evidence that older poststroke mice exhibited elevated intestinal inflammation and disruption in gut barriers critical in maintaining colonic integrity following stroke, including reduced expression of mucin and tight junction proteins. In addition, our data support the notion that the localized pro-inflammatory microenvironment driven by increased tumour necrosis factor-α production in the colon of older mice facilitates the translocation and dissemination of orally inoculated bacteria to the lung following stroke onset. Therefore, findings of this study demonstrate that exacerbated dysfunction of the intestinal barrier in advanced age promotes translocation of gut-derived bacteria and contributes to the increased risk to poststroke bacterial infection.
Keywords: aging; bacteria; colon; infection; stroke.
© 2019 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
None declared.
Figures





Similar articles
-
Dynamic Process of Secondary Pulmonary Infection in Mice With Intracerebral Hemorrhage.Front Immunol. 2021 Nov 19;12:767155. doi: 10.3389/fimmu.2021.767155. eCollection 2021. Front Immunol. 2021. PMID: 34868020 Free PMC article.
-
Ischemic stroke induces gut permeability and enhances bacterial translocation leading to sepsis in aged mice.Aging (Albany NY). 2016 May;8(5):1049-63. doi: 10.18632/aging.100952. Aging (Albany NY). 2016. PMID: 27115295 Free PMC article.
-
Translocation and dissemination of commensal bacteria in post-stroke infection.Nat Med. 2016 Nov;22(11):1277-1284. doi: 10.1038/nm.4194. Epub 2016 Oct 3. Nat Med. 2016. PMID: 27694934
-
Lung Imaging Reveals Stroke-Induced Impairment in Pulmonary Intravascular Neutrophil Function, a Response Exacerbated with Aging.J Immunol. 2022 Apr 15;208(8):2019-2028. doi: 10.4049/jimmunol.2100997. Epub 2022 Apr 1. J Immunol. 2022. PMID: 35365565
-
Enteric Pathogens and Their Toxin-Induced Disruption of the Intestinal Barrier through Alteration of Tight Junctions in Chickens.Toxins (Basel). 2017 Feb 10;9(2):60. doi: 10.3390/toxins9020060. Toxins (Basel). 2017. PMID: 28208612 Free PMC article. Review.
Cited by
-
Impact of aging and comorbidities on ischemic stroke outcomes in preclinical animal models: A translational perspective.Exp Neurol. 2021 Jan;335:113494. doi: 10.1016/j.expneurol.2020.113494. Epub 2020 Oct 7. Exp Neurol. 2021. PMID: 33035516 Free PMC article. Review.
-
Receptor-interacting protein kinase 2 (RIPK2) profoundly contributes to post-stroke neuroinflammation and behavioral deficits with microglia as unique perpetrators.J Neuroinflammation. 2023 Sep 30;20(1):221. doi: 10.1186/s12974-023-02907-6. J Neuroinflammation. 2023. PMID: 37777791 Free PMC article.
-
Immune regulation of the gut-brain axis and lung-brain axis involved in ischemic stroke.Neural Regen Res. 2024 Mar;19(3):519-528. doi: 10.4103/1673-5374.380869. Neural Regen Res. 2024. PMID: 37721279 Free PMC article. Review.
-
Gut Microbiota, Bacterial Translocation, and Stroke: Current Knowledge and Future Directions.Biomedicines. 2024 Dec 6;12(12):2781. doi: 10.3390/biomedicines12122781. Biomedicines. 2024. PMID: 39767686 Free PMC article. Review.
-
The Involvement of Immune Cells Between Ischemic Stroke and Gut Microbiota.Transl Stroke Res. 2024 Jun;15(3):498-517. doi: 10.1007/s12975-023-01151-7. Epub 2023 May 4. Transl Stroke Res. 2024. PMID: 37140808 Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

