Integrating Multiple Biomarkers to Increase Sensitivity for the Detection of Onchocerca volvulus Infection
- PMID: 31201416
- PMCID: PMC7213562
- DOI: 10.1093/infdis/jiz307
Integrating Multiple Biomarkers to Increase Sensitivity for the Detection of Onchocerca volvulus Infection
Abstract
Background: Serological assessments for human onchocerciasis are based on IgG4 reactivity against the OV-16 antigen, with sensitivities of 60-80%. We have previously identified 7 novel proteins that could improve serodiagnosis.
Methods: IgG4 responses to these 7 proteins were assessed by luciferase immunoprecipitation (LIPS) and enzyme-linked immunosorbent (ELISA) immunoassays.
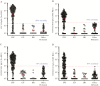
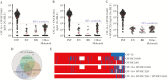
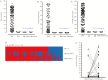
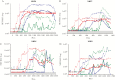
Results: OVOC10469 and OVOC3261 were identified as the most promising candidates by IgG4-based immunoassays with sensitivities of 53% for rOVOC10469 and 78% for rOVOC3261 while specificity for each was >99%. These 2 antigens in combination with OV-16 increased the sensitivity for patent infections to 94%. The kinetics of appearance of these IgG4 responses based on experimentally infected non-human primates indicated that they were microfilarial- driven. Further, the IgG4 responses to both OVOC10469 and OVOC3261 (as well as to OV-16) drop significantly (p<0.05) following successful treatment for onchocerciasis. A prototype lateral flow rapid diagnostic test to detect IgG4 to both Ov-16 and OVOC3261 was developed and tested demonstrating an overall 94% sensitivity.
Conclusion: The combined use of rOVOC3261 with OV-16 improved serologic assessment of O. volvulus infection, a current unmet need toward the goal of elimination of transmission of O. volvulus.
Keywords: OV-16; OVOC3261; biomarkers; diagnostics; onchocerciasis.
Published by Oxford University Press for the Infectious Diseases Society of America 2019.
Figures

Comment in
-
A Much-Needed Advance in the Diagnosis of River Blindness.J Infect Dis. 2020 May 11;221(11):1746-1748. doi: 10.1093/infdis/jiz309. J Infect Dis. 2020. PMID: 31201417 No abstract available.
Similar articles
-
Evaluation of an OV-16 IgG4 Enzyme-Linked Immunosorbent Assay in Humans and Its Application to Determine the Dynamics of Antibody Responses in a Non-Human Primate Model of Onchocerca volvulus Infection.Am J Trop Med Hyg. 2018 Oct;99(4):1041-1048. doi: 10.4269/ajtmh.18-0132. Am J Trop Med Hyg. 2018. PMID: 30062989 Free PMC article.
-
Characterizing Reactivity to Onchocerca volvulus Antigens in Multiplex Bead Assays.Am J Trop Med Hyg. 2017 Sep;97(3):666-672. doi: 10.4269/ajtmh.16-0519. Epub 2017 Jul 19. Am J Trop Med Hyg. 2017. PMID: 28722605 Free PMC article.
-
Evaluation of the Diagnostic Performance of Onchocerca volvulus Linear Epitopes in a Peptide Enzyme-Linked Immunosorbent Assay.Am J Trop Med Hyg. 2018 Mar;98(3):779-785. doi: 10.4269/ajtmh.17-0756. Epub 2018 Jan 4. Am J Trop Med Hyg. 2018. PMID: 29313477 Free PMC article.
-
Onchocerca volvulus infections in chimpanzees: an assessment of the requirements for vaccination trials.J Helminthol. 1994 Mar;68(1):3-6. doi: 10.1017/s0022149x00013390. J Helminthol. 1994. PMID: 8006381 Review. No abstract available.
-
CD4+-dependent immunity to Onchocerca volvulus third-stage larvae in humans and the mouse vaccination model: common ground and distinctions.Int J Parasitol. 2003 Sep 30;33(11):1161-71. doi: 10.1016/s0020-7519(03)00170-x. Int J Parasitol. 2003. PMID: 13678632 Review.
Cited by
-
House dust mite sensitization drives cross-reactive immune responses to homologous helminth proteins.PLoS Pathog. 2021 Mar 2;17(3):e1009337. doi: 10.1371/journal.ppat.1009337. eCollection 2021 Mar. PLoS Pathog. 2021. PMID: 33651853 Free PMC article.
-
Occurrence of Lymphatic Filariasis infection after 15 years of mass drug administration in two hotspot districts in the Upper East Region of Ghana.PLoS Negl Trop Dis. 2022 Aug 4;16(8):e0010129. doi: 10.1371/journal.pntd.0010129. eCollection 2022 Aug. PLoS Negl Trop Dis. 2022. PMID: 35926012 Free PMC article.
-
Post-Infectious Autoimmunity in the Central (CNS) and Peripheral (PNS) Nervous Systems: An African Perspective.Front Immunol. 2022 Mar 9;13:833548. doi: 10.3389/fimmu.2022.833548. eCollection 2022. Front Immunol. 2022. PMID: 35356001 Free PMC article. Review.
-
Comparison of Structural Changes in Nodding Syndrome and Other Epilepsies Associated With Onchocerca volvulus.Neurol Neuroimmunol Neuroinflamm. 2022 Dec 21;10(2):e200074. doi: 10.1212/NXI.0000000000200074. Print 2023 Mar. Neurol Neuroimmunol Neuroinflamm. 2022. PMID: 36543539 Free PMC article.
-
Molecular Mimicry Drives Locally Produced Autoantibodies in Subarachnoid Neurocysticercosis.Open Forum Infect Dis. 2025 May 8;12(5):ofaf276. doi: 10.1093/ofid/ofaf276. eCollection 2025 May. Open Forum Infect Dis. 2025. PMID: 40406372 Free PMC article.
References
-
- World Health Organization. Progress report on the elimination of human onchocerciasis, 2017–2018. Wkly Epidemiol Rec 2018; 47:633–48.
-
- World Health Organization. Progress towards eliminating onchocerciasis in the WHO Region of the Americas: elimination of transmission in the north-east focus of the Bolivarian Republic of Venezuela. Wkly Epidemiol Rec 2017; 92:617–23. - PubMed
-
- World Health Organization. Progress towards eliminating onchocerciasis in the WHO Region of the Americas: verification of elimination of transmission in Guatemala. Wkly Epidemiol Rec 2016; 91:501–5. - PubMed
-
- Ndyomugyenyi R, Lakwo T, Habomugisha P, Male B. Progress towards the elimination of onchocerciasis as a public-health problem in Uganda: opportunities, challenges and the way forward. Ann Trop Med Parasitol 2007; 101:323–33. - PubMed

