CRISPR/Cas9-mediated genome editing reveals 30 testis-enriched genes dispensable for male fertility in mice†
- PMID: 31201419
- PMCID: PMC6735960
- DOI: 10.1093/biolre/ioz103
CRISPR/Cas9-mediated genome editing reveals 30 testis-enriched genes dispensable for male fertility in mice†
Abstract
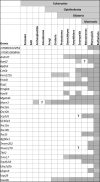
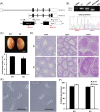
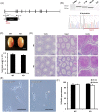
More than 1000 genes are predicted to be predominantly expressed in mouse testis, yet many of them remain unstudied in terms of their roles in spermatogenesis and sperm function and their essentiality in male reproduction. Since individually indispensable factors can provide important implications for the diagnosis of genetically related idiopathic male infertility and may serve as candidate targets for the development of nonhormonal male contraceptives, our laboratories continuously analyze the functions of testis-enriched genes in vivo by generating knockout mouse lines using the CRISPR/Cas9 system. The dispensability of genes in male reproduction is easily determined by examining the fecundity of knockout males. During our large-scale screening of essential factors, we knocked out 30 genes that have a strong bias of expression in the testis and are mostly conserved in mammalian species including human. Fertility tests reveal that the mutant males exhibited normal fecundity, suggesting these genes are individually dispensable for male reproduction. Since such functionally redundant genes are of diminished biological and clinical significance, we believe that it is crucial to disseminate this list of genes, along with their phenotypic information, to the scientific community to avoid unnecessary expenditure of time and research funds and duplication of efforts by other laboratories.
Keywords: CRISPR/Cas9; knockout mice; male infertility; spermatogenesis; testis expression.
© The Author(s) 2019. Published by Oxford University Press on behalf of Society for the Study of Reproduction.
Figures

References
-
- Dada R, Thilagavathi J, Venkatesh S, Esteves SC, Agarwal A. Genetic testing in male infertility. Open Reprod Sci J 2011; 3:42–56.
-
- Lotti F, Maggi M. Sexual dysfunction and male infertility. Nat Rev Urol 2018; 15:287–307. - PubMed
-
- Krausz C, Escamilla AR, Chianese C. Genetics of male infertility: from research to clinic. Reproduction 2015; 150:R159–R174. - PubMed
-
- Ghuman N, Ramalingam M. Male infertility. Obstet Gynaecol Reprod Med 2018; 28:7–14.
-
- Krausz C, Riera-Escamilla A. Genetics of male infertility. Nat Rev Urol 2018; 15:369–384. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

