Development and validation of a high performance liquid chromatography method to determine nevirapine in plasma in a resource-limited setting
- PMID: 31205870
- PMCID: PMC6556864
- DOI: 10.4102/ajlm.v8i1.880
Development and validation of a high performance liquid chromatography method to determine nevirapine in plasma in a resource-limited setting
Abstract
Background: There are several instances where nevirapine pharmacokinetic monitoring may be useful, such as in special populations or pharmacokinetic drug interaction studies that require the ascertainment of nevirapine pharmacokinetics in the sub-Saharan region.
Objectives: The main aim of this study was to produce a validated, sustainable and relevant nevirapine assay method that meets bio-analytical regulatory requirements.
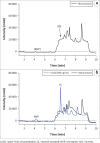
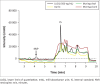
Methods: The developed method utilised a Waters 2795 Alliance high performance liquid chromatography system with a 2996 photo diode array detector, an Atlantis dC18 5 micron, 3.9 mm × 150 mm analytical column and a gradient flow rate of 1 mL/min. Ultraviolet detection data were collected from 210 nm to 400 nm, extracted at 260 nm, and processed for nevirapine and internal standard peak height responses.
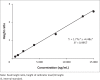
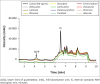
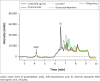
Results: The method proved to be linear (R2 0.995), precise (+1.92% - +9.69%) and accurate (-9.70% - 12.0%). Recovery for the analyte and internal standard was between 98.8% and 114%. The method showed good specificity as no interferences were caused by common African traditional medicines, anti-tuberculosis medications or other concomitant antiretrovirals nor endogenous components.
Conclusion: The method is reproducible, relevant to our setting and uses considerably low plasma volumes with preservation of some consumables, a desirable key factor in a resource-limited setting.
Keywords: high performance liquid chromatography; method development and validation; nevirapine determination.
Conflict of interest statement
The authors declare that they have no financial or personal relationships that may have inappropriately influenced them in writing this article.
Figures

References
-
- WHO Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV Infection: Recommendations for a public health approach. WHO guidelines approved by the guidelines review committee [homepage on the Internet]. Geneva; 2016. [updated 2016 Jun; 2018 Sept 19]. Available from: http://www.who.int/hiv/pub/arv/arv-2016/en/
-
- Niessens WMA, Correa CR. Introduction to LC-MS, interpretation of MS-MS mass spectra of drugs and pesticides [homepage on the Internet]. NJ, USA: John Wiley and Sons; 2017. [cited 2018 Apr 15]. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/9781119294269.ch1 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
