A model of guided cell self-organization for rapid and spontaneous formation of functional vessels
- PMID: 31206014
- PMCID: PMC6561743
- DOI: 10.1126/sciadv.aau6562
A model of guided cell self-organization for rapid and spontaneous formation of functional vessels
Abstract
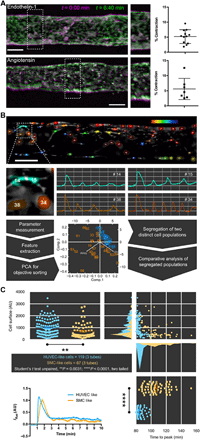
Most achievements to engineer blood vessels are based on multiple-step manipulations such as manual sheet rolling or sequential cell seeding followed by scaffold degradation. Here, we propose a one-step strategy using a microfluidic coextrusion device to produce mature functional blood vessels. A hollow alginate hydrogel tube is internally coated with extracellular matrix to direct the self-assembly of a mixture of endothelial cells (ECs) and smooth muscle cells (SMCs). The resulting vascular structure has the correct configuration of lumen, an inner lining of ECs, and outer sheath of SMCs. These "vesseloids" reach homeostasis within a day and exhibit the following properties expected for functional vessels (i) quiescence, (ii) perfusability, and (iii) contractility in response to vasoconstrictor agents. Together, these findings provide an original and simple strategy to generate functional artificial vessels and pave the way for further developments in vascular graft and tissue engineering and for deciphering the angiogenesis process.
Figures



References
-
- Cukierman E., Pankov R., Stevens D. R., Yamada K. M., Taking cell-matrix adhesions to the third dimension. Science 294, 1708–1712 (2001). - PubMed
-
- Zheng F., Fu F., Cheng Y., Wang C., Zhao Y., Gu Z., Organ-on-a-Chip Systems: Microengineering to biomimic living systems. Small 12, 2253–2282 (2016). - PubMed
-
- Bhatia S. N., Ingber D. E., Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

