Melatonin and agomelatine for preventing seasonal affective disorder
- PMID: 31206585
- PMCID: PMC6578031
- DOI: 10.1002/14651858.CD011271.pub3
Melatonin and agomelatine for preventing seasonal affective disorder
Abstract
Background: Seasonal affective disorder (SAD) is a seasonal pattern of recurrent major depressive episodes that most commonly starts in autumn or winter and remits in spring. The prevalence of SAD depends on latitude and ranges from 1.5% to 9%. The predictable seasonal aspect of SAD provides a promising opportunity for prevention in people who have a history of SAD. This is one of four reviews on the efficacy and safety of interventions to prevent SAD; we focus on agomelatine and melatonin as preventive interventions.
Objectives: To assess the efficacy and safety of agomelatine and melatonin (in comparison with each other, placebo, second-generation antidepressants, light therapy, psychological therapy or lifestyle interventions) in preventing SAD and improving person-centred outcomes among adults with a history of SAD.
Search methods: We searched Ovid MEDLINE (1950- ), Embase (1974- ), PsycINFO (1967- ) and the Cochrane Central Register of Controlled Trials (CENTRAL) to 19 June 2018. An earlier search of these databases was conducted via the Cochrane Common Mental Disorders Controlled Trial Register (CCMD-CTR) (all years to 11 August 2015). Furthermore, we searched the Cumulative Index to Nursing and Allied Health Literature, Web of Science, the Cochrane Library, the Allied and Complementary Medicine Database and international trial registers (to 19 June 2018). We also conducted a grey literature search and handsearched the reference lists of included studies and pertinent review articles.
Selection criteria: To examine efficacy, we included randomised controlled trials (RCTs) on adults with a history of winter-type SAD who were free of symptoms at the beginning of the study. For adverse events, we intended also to include non-randomised studies. We planned to include studies that compared agomelatine versus melatonin, or agomelatine or melatonin versus placebo, any second-generation antidepressant, light therapy, psychological therapies or lifestyle changes. We also intended to compare melatonin or agomelatine in combination with any of the comparator interventions mentioned above versus the same comparator intervention as monotherapy.
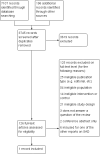
Data collection and analysis: Two review authors screened abstracts and full-text publications, abstracted data and assessed risk of bias of included studies independently. We intended to pool data in a meta-analysis using a random-effects model, but included only one study.
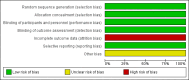
Main results: We identified 3745 citations through electronic searches and reviews of reference lists after deduplication of search results. We excluded 3619 records during title and abstract review and assessed 126 full-text papers for inclusion in the review. Only one study, providing data of 225 participants, met our eligibility criteria and compared agomelatine (25 mg/day) with placebo. We rated it as having high risk of attrition bias because nearly half of the participants left the study before completion. We rated the certainty of the evidence as very low for all outcomes, because of high risk of bias, indirectness, and imprecision.The main analysis based on data of 199 participants rendered an indeterminate result with wide confidence intervals (CIs) that may encompass both a relevant reduction as well as a relevant increase of SAD incidence by agomelatine (risk ratio (RR) 0.83, 95% CI 0.51 to 1.34; 199 participants; very low-certainty evidence). Also the severity of SAD may be similar in both groups at the end of the study with a mean SIGH-SAD (Structured Interview Guide for the Hamilton Depression Rating Scale, Seasonal Affective Disorders) score of 8.3 (standard deviation (SD) 9.4) in the agomelatine group and 10.1 (SD 10.6) in the placebo group (mean difference (MD) -1.80, 95% CI -4.58 to 0.98; 199 participants; very low-certainty evidence). The incidence of adverse events and serious adverse events may be similar in both groups. In the agomelatine group, 64 out of 112 participants experienced at least one adverse event, while 61 out of 113 did in the placebo group (RR 1.06, 95% CI 0.84 to 1.34; 225 participants; very low-certainty evidence). Three out of 112 patients experienced serious adverse events in the agomelatine group, compared to 4 out of 113 in the placebo group (RR 0.76, 95% CI 0.17 to 3.30; 225 participants; very low-certainty evidence).No data on quality of life or interpersonal functioning were reported. We did not identify any studies on melatonin.
Authors' conclusions: Given the uncertain evidence on agomelatine and the absence of studies on melatonin, no conclusion about efficacy and safety of agomelatine and melatonin for prevention of SAD can currently be drawn. The decision for or against initiating preventive treatment of SAD and the treatment selected should consider patient preferences and reflect on the evidence base of all available treatment options.
Conflict of interest statement
Barbara Nussbaumer‐Streit ‐ no conflict of interest
Amy Greenblatt ‐ no conflict of interest
Angela Kaminski‐Hartenthaler ‐ no conflict of interest
Megan G Van Noord ‐ no conflict of interest
Catherine A Forneris ‐ no conflict of interest
Laura C Morgan ‐ no conflict of interest
Bradley N Gaynes ‐ no conflict of interest
Jörg Wipplinger ‐ no conflict of interest
Linda J Lux ‐ no conflict of interest
Dietmar Winkler received lecture fees/authorship honoraria from Angelini, Lundbeck, and Medizin Medien Austria
Gerald Gartlehner ‐ no conflict of interest
Figures


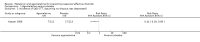

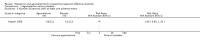
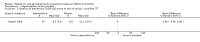
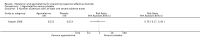

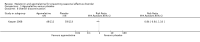
Update of
-
Melatonin and agomelatine for preventing seasonal affective disorder.Cochrane Database Syst Rev. 2015 Nov 11;(11):CD011271. doi: 10.1002/14651858.CD011271.pub2. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2019 Jun 17;6:CD011271. doi: 10.1002/14651858.CD011271.pub3. PMID: 26560173 Updated.
Similar articles
-
Psychological therapies for preventing seasonal affective disorder.Cochrane Database Syst Rev. 2019 May 24;5(5):CD011270. doi: 10.1002/14651858.CD011270.pub3. Cochrane Database Syst Rev. 2019. PMID: 31124141 Free PMC article.
-
Light therapy for preventing seasonal affective disorder.Cochrane Database Syst Rev. 2019 Mar 18;3(3):CD011269. doi: 10.1002/14651858.CD011269.pub3. Cochrane Database Syst Rev. 2019. PMID: 30883670 Free PMC article.
-
Melatonin and agomelatine for preventing seasonal affective disorder.Cochrane Database Syst Rev. 2015 Nov 11;(11):CD011271. doi: 10.1002/14651858.CD011271.pub2. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2019 Jun 17;6:CD011271. doi: 10.1002/14651858.CD011271.pub3. PMID: 26560173 Updated.
-
Second-generation antidepressants for preventing seasonal affective disorder in adults.Cochrane Database Syst Rev. 2019 Mar 18;3(3):CD011268. doi: 10.1002/14651858.CD011268.pub3. Cochrane Database Syst Rev. 2019. PMID: 30883669 Free PMC article.
-
Light therapy for preventing seasonal affective disorder.Cochrane Database Syst Rev. 2015 Nov 8;(11):CD011269. doi: 10.1002/14651858.CD011269.pub2. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2019 Mar 18;3:CD011269. doi: 10.1002/14651858.CD011269.pub3. PMID: 26558494 Updated.
Cited by
-
Agomelatine prevents angiotensin II-induced endothelial and mononuclear cell adhesion.Aging (Albany NY). 2021 Jul 22;13(14):18515-18526. doi: 10.18632/aging.203299. Epub 2021 Jul 22. Aging (Albany NY). 2021. PMID: 34292876 Free PMC article.
-
The darkness and the light: diurnal rodent models for seasonal affective disorder.Dis Model Mech. 2021 Jan 26;14(1):dmm047217. doi: 10.1242/dmm.047217. Dis Model Mech. 2021. PMID: 33735098 Free PMC article. Review.
-
Melatonin and Depression: A Translational Perspective From Animal Models to Clinical Studies.Front Psychiatry. 2021 Apr 8;12:638981. doi: 10.3389/fpsyt.2021.638981. eCollection 2021. Front Psychiatry. 2021. PMID: 33897495 Free PMC article. Review.
-
Predictability of Seasonal Mood Fluctuations Based on Self-Report Questionnaires and EEG Biomarkers in a Non-clinical Sample.Front Psychiatry. 2022 Apr 8;13:870079. doi: 10.3389/fpsyt.2022.870079. eCollection 2022. Front Psychiatry. 2022. PMID: 35463521 Free PMC article.
-
Self-care for anxiety and depression: a comparison of evidence from Cochrane reviews and practice to inform decision-making and priority-setting.BMC Complement Med Ther. 2020 Aug 10;20(1):247. doi: 10.1186/s12906-020-03038-8. BMC Complement Med Ther. 2020. PMID: 32778171 Free PMC article.
References
References to studies included in this review
Kasper 2008 {unpublished data only}
-
- European Medicines Agency. CHMP Assessment Report for Thymax. [Procedure No. EMEA/H/C/000916]. www.ema.europa.eu/en/documents/assessment‐report/thymanax‐epar‐public‐as... 2008; Vol. accessed 11 June 2019.
-
- Kasper S, Lam R, Partonen TT, Moller HJ, Boer J, Magnusson A, et al. Efficacy and safety of S 20098 (25 mg) in the prevention of recurrence, in patients with a seasonal affective disorder, treated once a day for 1 year after an open period of at least 18 weeks. A randomised, double‐blind, placebo‐controlled, parallel group study [CL3‐20098‐037]. Institut de Recherches Internationales Servier (I.R.I.S.) 2008; Vol. 1:1‐131 [Confidential].
References to studies excluded from this review
Fleer 2014 {published data only}
-
- Fleer J, Schroevers M, Panjer V, Geerts E, Meesters Y. Mindfulness‐based cognitive therapy for seasonal affective disorder: A pilot study. Journal of Affective Disorder 2014;168:205‐9. - PubMed
Goodwin 2009 {published data only}
-
- Goodwin GM, Emsley R, Rembry S, Rouillon F. Agomelatine prevents relapse in patients with major depressive disorder without evidence of a discontinuation syndrome: a 24‐week randomized, double‐blind, placebo‐controlled trial. Journal of Clinical Psychiatry 2009;70:1128‐37. - PubMed
Leppämäki 2003 {published data only}
-
- Leppämäki S, Partonen T, Vakkuri O, Lönnqvist J, Partinen M, Laudon M. Effect of controlled‐release melatonin on sleep quality, mood, and quality of life in subjects with seasonal or weather‐associated changes in mood and behaviour. European Neuropsychopharmacology 2003; Vol. 13:137‐45. - PubMed
Meesters 1999 {published data only}
-
- Meesters Y, Beersma DG, Bouhuys AL, Hoofdakker RH. Prophylactic treatment of seasonal affective disorder (SAD) by using light visors: bright white or infrared light?. Biological Psychiatry 1999;46:239‐46. - PubMed
Modell 2005 {published data only}
-
- Modell JG, Rosenthal NE, Harriett AE, Krishen A, Asgharian A, Foster VJ, et al. Seasonal affective disorder and its prevention by anticipatory treatment with bupropion XL. Biological Psychiatry 2005;58:658‐67. - PubMed
WELL 100006 {published data only}
-
- WELL 100006. A 7‐month, multicenter, randomized, double‐blind, placebo‐controlled comparison of 150‐300mg/day of extended‐release bupropion hydrochloride (WELLBUTRIN XL) and placebo for the prevention of seasonal affective disorder in subjects with a history of seasonal affective disorder followed by an 8‐week observational follow‐up phase. GSK ‐ Clinical Study Register [www.gsk‐clinicalstudyregister.com] 2003.
WELL AK130936 {published data only}
-
- WELL AK130936. A 7‐month, multicenter, randomized, double‐blind, placebo‐controlled comparison of 150‐300mg/day of extended‐release bupropion hydrochloride (WELLBUTRIN XL) and placebo for the prevention of seasonal affective disorder in subjects with a history of seasonal affective disorder followed by an 8‐week observational follow‐up study. GSK ‐ Clinical Study Register [www.gsk‐clinicalstudyregister.com] 2004.
WELL AKA130930 {published data only}
-
- WELL AKA130930. A 7‐month, multicenter, randomized, double‐blind, placebo‐controlled comparison of 150‐300mg/day of extended‐release bupropion hydrochloride (WELLBUTRIN XL) and placebo for the prevention of seasonal affective disorder in subjects with a history of seasonal affective disorder followed by an 8‐week observational follow‐up phase. GSK ‐ Clinical Study Register [www.gsk‐clinicalstudyregister.com] 2003.
Wirz‐Justice 1990 {published data only}
-
- Wirz‐Justice A, Graw P, Krauchi K, Gisin B, Arendt J, Aldhous M, et al. Morning or night‐time melatonin is ineffective in seasonal affective disorder. Journal of Psychiatric Research 1990;24:129‐37. - PubMed
Additional references
APA 1980
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd Edition. Washington, DC: American Psychiatric Association, 1980.
APA 1987
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd Edition. Washington, DC: American Psychiatric Association, 1987.
APA 2000
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th Edition. Washington, DC: American Psychiatric Association, 2000.
APA 2013
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th Edition. Washington, DC: American Psychiatric Association, 2013.
Begg 1994
-
- Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994;50:1088‐101. - PubMed
Byrne 2008
-
- Byrne B, Brainard GC. Seasonal affective disorder and light therapy. Sleep Medicine Clinics 2008;3(2):307‐15.
Ciarleglio 2011
-
- Ciarleglio CM, Resuehr HE, McMahon DG. Interactions of the serotonin and circadian systems: nature and nurture in rhythms and blues. Neuroscience 2011;197:8‐16. - PubMed
Egger 1997
EMA 2009
-
- European Medicines Agency. CHMP Assessment Report for Thymanax. Procedure No. EMEA/H/C/000916 2009:1‐66.
EndNote X8 2016 [Computer program]
-
- Clarivate Analytics. EndNote X8. Version accessed prior to 13 May 2019. Philadelphia: Clarivate Analytics, 2016.
Forneris 2019
Gartlehner 2019
GRADEpro GDT 2015 [Computer program]
-
- McMaster University (Developed by Evidence Prime). GRADEpro GDT. Hamilton (ON): McMaster University (Developed by Evidence Prime), 2015.
Guyatt 2011
-
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011;64(4):383‐94. - PubMed
Hamilton 1960
Hansen 2009
Hickie 2011
-
- Hickie IB, Rogers NL. Novel melatonin‐based therapies: potential advances in the treatment of major depression. Lancet 2011;378(9791):621‐31. - PubMed
Higgins 2011
-
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Kasper 2010
-
- Kasper S, Hajak G, Wulff K, Hoogendijk WJ, Montejo AL, Smeraldi E, et al. Efficacy of the novel antidepressant agomelatine on the circadian rest‐activity cycle and depressive and anxiety symptoms in patients with major depressive disorder: a randomized, double‐blind comparison with sertraline. Journal of Clinical Psychiatry 2010;71(2):109‐20. - PubMed
Lam 2006
-
- Lam RW, Levitt AJ, Levitan RD, Enns MW, Morehouse R, Michalak EE, et al. The Can‐SAD study: a randomized controlled trial of the effectiveness of light therapy and fluoxetine in patients with winter seasonal affective disorder. American Journal of Psychiatry 2006;163(5):805‐12. - PubMed
Leon 1999
-
- Leon AC, Solomon DA, Mueller TI, Turvey CL, Endicott J, Keller MB. The Range of Impaired Functioning Tool (LIFE‐RIFT): a brief measure of functional impairment. Psychological Medicine 1999;29(4):869‐78. - PubMed
Levitan 2007
Lewy 1988
-
- Lewy AJ, Sack RL, Singer CM, White DM, Hoban TM. Winter depression and the phase‐shift hypothesis for bright light's therapeutic effects: history, theory, and experimental evidence. Journal of Biological Rhythms 1988;3(2):121‐34. - PubMed
Lewy 2006
-
- Lewy AJ, Emens J, Jackman A, Yuhas K. Circadian uses of melatonin in humans. Chronobiology International 2006;23(1‐2):403‐12. - PubMed
Magnusson 2005
-
- Magnusson A, Partonen T. The diagnosis, symptomatology, and epidemiology of seasonal affective disorder. CNS Spectrums 2005;10(8):625‐34. - PubMed
Moher 2009
Neumeister 2001
-
- Neumeister A, Konstantinidis A, Praschak‐Rieder N, Willeit M, Hilger E, Stastny J, et al. Monoaminergic function in the pathogenesis of seasonal affective disorder. International Journal of Neuropsychopharmacology 2001;4:409‐20. - PubMed
Nussbaumer‐Streit 2017
Nussbaumer‐Streit 2018
Nussbaumer‐Streit 2019
Partonen 1998
-
- Partonen T, Lonnqvist J. Seasonal affective disorder. Lancet 1998;352:1369‐74. - PubMed
Pjrek 2007
-
- Pjrek E, Winkler D, Konstantinidis A, Willeit M, Praschak‐Rieder N, Kasper S. Agomelatine in the treatment of seasonal affective disorder. Psychopharmacology 2007;190(4):575‐9. - PubMed
Review Manager 2014 [Computer program]
-
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Rodin 1997
-
- Rodin I, Thompson C. Seasonal affective disorder. Advances in Psychiatric Treatment 1997;3:352‐9.
Rosen 1990
-
- Rosen LN, Targum SD, Terman M, Bryant MJ, Hoffman H, Kasper SF. Prevalence of seasonal affective disorder at four latitudes. Psychiatry Research 1990;31:131‐44. - PubMed
Rosenthal 1979
-
- Rosenthal R. The "file‐drawer problem" and tolerance for null results. Psychological Bulletin 1979;86:638‐41.
Rosenthal 1984
-
- Rosenthal NE, Sack DA, Gillin JC, Lewy AJ, Goodwin FK, Davenport Y, et al. Seasonal affective disorder. A description of the syndrome and preliminary findings with light therapy. Archives of General Psychiatry 1984;41(1):72‐80. - PubMed
Schwartz 1996
-
- Schwartz PJ, Brown C, Wehr TA, Rosenthal NE. Winter seasonal affective disorder: a follow‐up study of the first 59 patients of the National Institute of Mental Health Seasonal Studies Program. American Journal of Psychiatry 1996;153(8):1028‐36. - PubMed
Segal 2010
-
- Segal DL. Diagnostic and Statistical Manual of Mental Disorders (DSM‐IV‐TR). The Corsini Encyclopedia of Psychology. John Wiley & Sons, Inc, 2010:495‐7.
Sohn 2005
-
- Sohn CH, Lam RW. Update on the biology of seasonal affective disorder. CNS Spectrums 2005;10(8):635‐46. - PubMed
Srinivasan 2012
-
- Srinivasan V, Berardis D, Shillcutt SD, Brzezinski A. Role of melatonin in mood disorders and the antidepressant effects of agomelatine. Expert Opinion on Investigational Drugs 2012;21(10):1503‐22. - PubMed
Terman 2005a
-
- Terman M, Terman JS. Light therapy for seasonal and nonseasonal depression: efficacy, protocol, safety, and side effects. CNS spectrums 2005;10(8):647‐63; quiz 672. - PubMed
Thaler 2010
Ware 1992
-
- Ware JE Jr, Sherbourne CD. The MOS 36‐item short‐form health survey (SF‐36). I. Conceptual framework and item selection. Medical Care 1992;30(6):473‐83. - PubMed
Wells 2009
-
- Wells GA, Shea B, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta‐analyses. www.ohri.ca/programs/clinical_epidemiology/oxford.htm 2009.
Westrin 2007
-
- Westrin A, Lam RW. Long‐term and preventative treatment for seasonal affective disorder. CNS Drugs 2007;21(11):901‐9. - PubMed
Williams 2002
-
- Williams JBW, Link MJ, Rosenthal NE, Terman M. Structured Interview Guide for the Hamilton Depression Rating Scale ‐ Seasonal Affective Disorder version (SIGH‐SAD). New York: New York State Psychiatric Institute, 2002.
References to other published versions of this review
Kaminski‐Hartenthaler 2015
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

