Adipose Tissue-Derived Signatures for Obesity and Type 2 Diabetes: Adipokines, Batokines and MicroRNAs
- PMID: 31208019
- PMCID: PMC6617388
- DOI: 10.3390/jcm8060854
Adipose Tissue-Derived Signatures for Obesity and Type 2 Diabetes: Adipokines, Batokines and MicroRNAs
Abstract
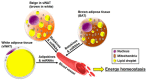
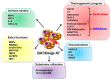
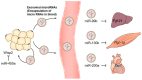
: Obesity is one of the main risk factors for type 2 diabetes mellitus (T2DM). It is closely related to metabolic disturbances in the adipose tissue that primarily functions as a fat reservoir. For this reason, adipose tissue is considered as the primary site for initiation and aggravation of obesity and T2DM. As a key endocrine organ, the adipose tissue communicates with other organs, such as the brain, liver, muscle, and pancreas, for the maintenance of energy homeostasis. Two different types of adipose tissues-the white adipose tissue (WAT) and brown adipose tissue (BAT)-secrete bioactive peptides and proteins, known as "adipokines" and "batokines," respectively. Some of them have beneficial anti-inflammatory effects, while others have harmful inflammatory effects. Recently, "exosomal microRNAs (miRNAs)" were identified as novel adipokines, as adipose tissue-derived exosomal miRNAs can affect other organs. In the present review, we discuss the role of adipose-derived secretory factors-adipokines, batokines, and exosomal miRNA-in obesity and T2DM. It will provide new insights into the pathophysiological mechanisms involved in disturbances of adipose-derived factors and will support the development of adipose-derived factors as potential therapeutic targets for obesity and T2DM.
Keywords: adipokines; batokines; exosomal miRNAs; obesity; potential therapeutic targets; type 2 diabetes mellitus.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Boden G., Homko C., Barrero C.A., Stein T.P., Chen X., Cheung P., Fecchio C., Koller S., Merali S. Excessive caloric intake acutely causes oxidative stress, GLUT4 carbonylation, and insulin resistance in healthy men. Sci. Transl. Med. 2015;7:304re7. doi: 10.1126/scitranslmed.aac4765. - DOI - PMC - PubMed
-
- LeRoith D., Novosyadlyy R., Gallagher E.J., Lann D., Vijayakumar A., Yakar S. Obesity and type 2 diabetes are associated with an increased risk of developing cancer and a worse prognosis; epidemiological and mechanistic evidence. Exp. Clin. Endocrinol. Diabetes. 2008;116(Suppl. 1):S4–S6. doi: 10.1055/s-2008-1081488. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

