Unique Regulation of Na-K-ATPase during Growth and Maturation of Intestinal Epithelial Cells
- PMID: 31208048
- PMCID: PMC6628168
- DOI: 10.3390/cells8060593
Unique Regulation of Na-K-ATPase during Growth and Maturation of Intestinal Epithelial Cells
Abstract
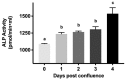
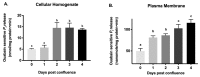
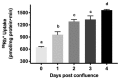
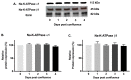
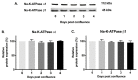
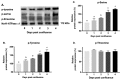
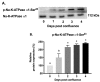
Na-K-ATPase on the basolateral membrane provides the favorable transcellular Na gradient for the proper functioning of Na-dependent nutrient co-transporters on the brush border membrane (BBM) of enterocytes. As cells mature from crypts to villus, Na-K-ATPase activity doubles, to accommodate for the increased BBM Na-dependent nutrient absorption. However, the mechanism of increased Na-K-ATPase activity during the maturation of enterocytes is not known. Therefore, this study aimed to determine the mechanisms involved in the functional transition of Na-K-ATPase during the maturation of crypts to villus cells. Na-K-ATPase activity gradually increased as IEC-18 cells matured in vitro from day 0 (crypts) through day 4 (villus) of post-confluence. mRNA abundance and Western blot studies showed no change in the levels of Na-K-ATPase subunits α1 and β1 from 0 to 4 days post-confluent cells. However, Na-K-ATPase α1 phosphorylation levels on serine and tyrosine, but not threonine, residues gradually increased. These data indicate that as enterocytes mature from crypt-like to villus-like in culture, the functional activity of Na-K-ATPase increases secondary to altered affinity of the α1 subunit to extracellular K+, in order to accommodate the functional preference of the intestinal cell type. This altered affinity is likely due to increased phosphorylation of the α1 subunit, specifically at serine and tyrosine residues.
Keywords: Na-dependent nutrient co-transport; Na/K-ATPase; cell maturation; crypt cells; intestinal absorption; villus cells.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
Mechanism of Na-K-ATPase Inhibition by PGE2 in Intestinal Epithelial Cells.Cells. 2021 Mar 29;10(4):752. doi: 10.3390/cells10040752. Cells. 2021. PMID: 33805551 Free PMC article.
-
Molecular mechanism of regulation of villus cell Na-K-ATPase in the chronically inflamed mammalian small intestine.Biochim Biophys Acta. 2015 Feb;1848(2):702-11. doi: 10.1016/j.bbamem.2014.11.005. Epub 2014 Nov 22. Biochim Biophys Acta. 2015. PMID: 25462166
-
Chronic and selective inhibition of basolateral membrane Na-K-ATPase uniquely regulates brush border membrane Na absorption in intestinal epithelial cells.Am J Physiol Cell Physiol. 2015 Apr 15;308(8):C650-6. doi: 10.1152/ajpcell.00355.2014. Epub 2015 Feb 4. Am J Physiol Cell Physiol. 2015. PMID: 25652450 Free PMC article.
-
Short-term aldosterone action on Na,K-ATPase surface expression: role of aldosterone-induced SGK1?Ann N Y Acad Sci. 2003 Apr;986:554-61. doi: 10.1111/j.1749-6632.2003.tb07253.x. Ann N Y Acad Sci. 2003. PMID: 12763889 Review.
-
Transcriptional regulators of Na,K-ATPase subunits.Front Cell Dev Biol. 2015 Oct 26;3:66. doi: 10.3389/fcell.2015.00066. eCollection 2015. Front Cell Dev Biol. 2015. PMID: 26579519 Free PMC article. Review.
Cited by
-
Spondias pinnata bark extract- an ameliorator of inflammatory derangement in etoposide induced mucositis: An experimental approach.Vet World. 2021 Jul;14(7):1822-1828. doi: 10.14202/vetworld.2021.1822-1828. Epub 2021 Jul 15. Vet World. 2021. PMID: 34475704 Free PMC article.
-
Mechanism of Na-K-ATPase Inhibition by PGE2 in Intestinal Epithelial Cells.Cells. 2021 Mar 29;10(4):752. doi: 10.3390/cells10040752. Cells. 2021. PMID: 33805551 Free PMC article.
-
The Role of Metal Oxide Nanoparticles, Escherichia coli, and Lactobacillus rhamnosus on Small Intestinal Enzyme Activity.Environ Sci Nano. 2020 Dec 1;7(12):3940-3964. doi: 10.1039/d0en01001d. Epub 2020 Nov 9. Environ Sci Nano. 2020. PMID: 33815806 Free PMC article.
-
Moderate Alcohol Consumption Uniquely Regulates Sodium-Dependent Glucose Co-Transport in Rat Intestinal Epithelial Cells In Vitro and In Vivo.J Nutr. 2020 Apr 1;150(4):747-755. doi: 10.1093/jn/nxz277. J Nutr. 2020. PMID: 31769840 Free PMC article.
-
Proliferation and Differentiation of Intestinal Caco-2 Cells Are Maintained in Culture with Human Platelet Lysate Instead of Fetal Calf Serum.Cells. 2021 Nov 5;10(11):3038. doi: 10.3390/cells10113038. Cells. 2021. PMID: 34831260 Free PMC article.
References
-
- Babyatsky M.W., Podolsky D.K. Growth and development of the gastrointestinal tract. In: Yamada T., editor. Textbook of Gastroenterology. Lippincott; Philadelphia, PA, USA: 1999. pp. 547–584.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

