Standardised imaging pipeline for phenotyping mouse laterality defects and associated heart malformations, at multiple scales and multiple stages
- PMID: 31208960
- PMCID: PMC6679386
- DOI: 10.1242/dmm.038356
Standardised imaging pipeline for phenotyping mouse laterality defects and associated heart malformations, at multiple scales and multiple stages
Abstract
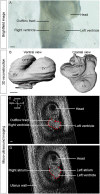
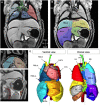
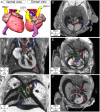
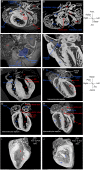
Laterality defects are developmental disorders resulting from aberrant left/right patterning. In the most severe cases, such as in heterotaxy, they are associated with complex malformations of the heart. Advances in understanding the underlying physiopathological mechanisms have been hindered by the lack of a standardised and exhaustive procedure in mouse models for phenotyping left/right asymmetries of all visceral organs. Here, we have developed a multimodality imaging pipeline, which combines non-invasive micro-ultrasound imaging, micro-computed tomography (micro-CT) and high-resolution episcopic microscopy (HREM) to acquire 3D images at multiple stages of development and at multiple scales. On the basis of the position in the uterine horns, we track in a single individual, the progression of organ asymmetry, the situs of all visceral organs in the thoracic or abdominal environment, and the fine anatomical left/right asymmetries of cardiac segments. We provide reference anatomical images and organ reconstructions in the mouse, and discuss differences with humans. This standardised pipeline, which we validated in a mouse model of heterotaxy, offers a fast and easy-to-implement framework. The extensive 3D phenotyping of organ asymmetry in the mouse uses the clinical nomenclature for direct comparison with patient phenotypes. It is compatible with automated and quantitative image analyses, which is essential to compare mutant phenotypes with incomplete penetrance and to gain mechanistic insight into laterality defects.
Keywords: 3D imaging; Congenital heart defects; Heterotaxy; Laterality defects; Left-right asymmetry; Situs.
© 2019. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures

Similar articles
-
Left-right patterning in congenital heart disease beyond heterotaxy.Am J Med Genet C Semin Med Genet. 2020 Mar;184(1):90-96. doi: 10.1002/ajmg.c.31768. Epub 2020 Jan 30. Am J Med Genet C Semin Med Genet. 2020. PMID: 31999049 Free PMC article. Review.
-
Phenotyping cardiac and structural birth defects in fetal and newborn mice.Birth Defects Res. 2017 Jun 1;109(10):778-790. doi: 10.1002/bdr2.1048. Epub 2017 May 22. Birth Defects Res. 2017. PMID: 28544620 Free PMC article. Review.
-
Imaging techniques for visualizing and phenotyping congenital heart defects in murine models.Birth Defects Res C Embryo Today. 2013 Jun;99(2):93-105. doi: 10.1002/bdrc.21037. Birth Defects Res C Embryo Today. 2013. PMID: 23897594 Free PMC article. Review.
-
The nomenclature, definition and classification of cardiac structures in the setting of heterotaxy.Cardiol Young. 2007 Sep;17 Suppl 2:1-28. doi: 10.1017/S1047951107001138. Cardiol Young. 2007. PMID: 18039396 Review.
-
Mouse model of heterotaxy with single ventricle spectrum of cardiac anomalies.Pediatr Res. 2008 Jan;63(1):9-14. doi: 10.1203/PDR.0b013e31815b6926. Pediatr Res. 2008. PMID: 18043505
Cited by
-
The venous system of E14.5 mouse embryos-reference data and examples for diagnosing malformations in embryos with gene deletions.J Anat. 2022 Jan;240(1):11-22. doi: 10.1111/joa.13536. Epub 2021 Aug 25. J Anat. 2022. PMID: 34435363 Free PMC article.
-
Label-free optical imaging in developmental biology [Invited].Biomed Opt Express. 2020 Mar 13;11(4):2017-2040. doi: 10.1364/BOE.381359. eCollection 2020 Apr 1. Biomed Opt Express. 2020. PMID: 32341864 Free PMC article. Review.
-
morphoHeart: A quantitative tool for integrated 3D morphometric analyses of heart and ECM during embryonic development.PLoS Biol. 2025 Jan 29;23(1):e3002995. doi: 10.1371/journal.pbio.3002995. eCollection 2025 Jan. PLoS Biol. 2025. PMID: 39879226 Free PMC article.
-
Mouse embryo phenotyping using X-ray microCT.Front Cell Dev Biol. 2022 Sep 16;10:949184. doi: 10.3389/fcell.2022.949184. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36187491 Free PMC article. Review.
-
Cardiac multi-scale investigation of the right and left ventricle ex vivo: a review.Cardiovasc Diagn Ther. 2020 Oct;10(5):1701-1717. doi: 10.21037/cdt-20-269. Cardiovasc Diagn Ther. 2020. PMID: 33224784 Free PMC article. Review.
References
-
- Couinaud C. (1957). Le foie; études anatomiques et chirurgicales. Paris: Masson.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

