Design and application of α-ketothioesters as 1,2-dicarbonyl-forming reagents
- PMID: 31209228
- PMCID: PMC6572800
- DOI: 10.1038/s41467-019-10651-w
Design and application of α-ketothioesters as 1,2-dicarbonyl-forming reagents
Abstract
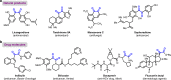
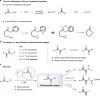
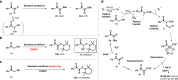
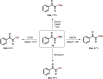
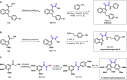
The 1,2-dicarbonyl motif is vital to biomolecules, especially natural products and pharmaceuticals. Conventionally, 1,2-dicarbonyl compounds are prepared via an α-keto acyl chloride. Based on the methods used in nature, a transition-metal-free approach for the synthesis of an α-ketothioester reagent via the combination of an α-hydroxyl ketone, elemental sulfur and a benzyl halide is reported. Mechanistic studies demonstrate that the trisulfur radical anion and the α-carbon radical of the α-hydroxy ketone are involved in this transformation. The dicarbonylation of a broad range of amines and amino acids, and importantly, cross couplings with aryl borates to construct dicarbonyl-carbon bonds are realized under mild conditions by employing this stable and convenient α-ketothioester as a 1,2-dicarbonyl reagent. The dicarbonyl-containing drug indibulin and the natural product polyandrocarpamide C, which possess multiple heteroatoms and active hydrogen functional groups, can be efficiently prepared using the designed 1,2-dicarbonyl reagent.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Bettòlo GBM, Casinovi CG, Galeffi C. A new class of quinones: sesquiterpenoid quinones of Mansonia Altissima. Tetrahedron Lett. 1965;52:4857–4864. doi: 10.1016/S0040-4039(01)89048-1. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

