Pentraxin 3 deficiency exacerbates lipopolysaccharide-induced inflammation in adipose tissue
- PMID: 31209269
- PMCID: PMC6997122
- DOI: 10.1038/s41366-019-0402-4
Pentraxin 3 deficiency exacerbates lipopolysaccharide-induced inflammation in adipose tissue
Abstract
Background/objectives: Pentraxin 3 (PTX3) has been characterized as a soluble and multifunctional pattern recognition protein in the regulation of innate immune response. However, little is known about its role in adipose tissue inflammation and obesity. Herein, we investigated the role of PTX3 in the regulation of lipopolysaccharide (LPS)-induced inflammation in adipocytes and adipose tissue, as well as high-fat diet (HFD)-induced metabolic inflammation in obesity.
Methods: Ptx3 knockdown 3T3-L1 Cells were generated using shRNA for Ptx3 gene and treated with different inflammatory stimuli. For the in vivo studies, Ptx3 knockout mice were treated with 0.3 mg/kg of LPS for 6 h. Adipose tissues were collected for gene and protein expression by qPCR and western blotting, respectively. Ptx3 knockout mice were fed with HFD for 12 week since 6 week of age.
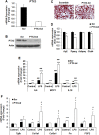
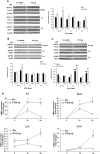
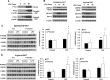
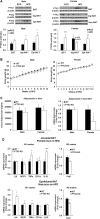
Results: We observed that the expression of PTX3 in adipose tissue and serum PTX3 were markedly increased in response to LPS administration. Knocking down Ptx3 in 3T3-L1 cells reduced adipogenesis and caused a more profound and sustained upregulation of proinflammatory gene expression and signaling pathway activation during LPS-stimulated inflammation in 3T3-L1 adipocytes. In vivo studies showed that PTX3 deficiency significantly exacerbated the LPS-induced upregulation of inflammatory genes and downregulation of adipogeneic genes in visceral and subcutaneous adipose tissue of mice. Accordingly, LPS stimulation elicited increased activation of nuclear factor-κB (NF-κB) and p44/42 MAPK (Erk1/2) signaling pathways in visceral and subcutaneous adipose tissue. The expression of PTX3 in adipose tissue was also induced by HFD, and PTX3 deficiency led to the upregulation of proinflammatory genes in visceral adipose tissue of HFD-induced obese mice.
Conclusions: Our results suggest a protective role of PTX3 in LPS- and HFD-induced sustained inflammation in adipose tissue.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Pentraxin 3 deficiency protects from the metabolic inflammation associated to diet-induced obesity.Cardiovasc Res. 2019 Nov 1;115(13):1861-1872. doi: 10.1093/cvr/cvz068. Cardiovasc Res. 2019. PMID: 30859179
-
Pentraxin 3 Regulates miR-21 Expression and Secretion in Brown Adipocytes During Lipopolysaccharide-Induced Inflammation.Obesity (Silver Spring). 2020 Feb;28(2):323-332. doi: 10.1002/oby.22701. Epub 2020 Jan 3. Obesity (Silver Spring). 2020. PMID: 31898407
-
Elevated Pentraxin 3 in Obese Adipose Tissue Promotes Adipogenic Differentiation by Activating Neuropeptide Y Signaling.Front Immunol. 2018 Jul 30;9:1790. doi: 10.3389/fimmu.2018.01790. eCollection 2018. Front Immunol. 2018. PMID: 30105036 Free PMC article.
-
Silencing of PTX3 alleviates LPS-induced inflammatory pain by regulating TLR4/NF-κB signaling pathway in mice.Biosci Rep. 2020 Feb 28;40(2):BSR20194208. doi: 10.1042/BSR20194208. Biosci Rep. 2020. PMID: 31957804 Free PMC article.
-
Role of microbiota-derived lipopolysaccharide in adipose tissue inflammation, adipocyte size and pyroptosis during obesity.Nutr Res Rev. 2018 Dec;31(2):153-163. doi: 10.1017/S0954422417000269. Epub 2018 Jan 24. Nutr Res Rev. 2018. PMID: 29362018 Review.
Cited by
-
Vesicles Shed by Pathological Murine Adipocytes Spread Pathology: Characterization and Functional Role of Insulin Resistant/Hypertrophied Adiposomes.Int J Mol Sci. 2020 Mar 24;21(6):2252. doi: 10.3390/ijms21062252. Int J Mol Sci. 2020. PMID: 32214011 Free PMC article.
-
Adipose tissue depot specific expression and regulation of fibrosis-related genes and proteins in experimental obesity.Mamm Genome. 2024 Mar;35(1):13-30. doi: 10.1007/s00335-023-10022-3. Epub 2023 Oct 26. Mamm Genome. 2024. PMID: 37884762 Free PMC article.
-
Loss of Uncoupling Protein 1 Expression in the Subcutaneous Adipose Tissue Predicts Childhood Obesity.Int J Mol Sci. 2023 Nov 24;24(23):16706. doi: 10.3390/ijms242316706. Int J Mol Sci. 2023. PMID: 38069028 Free PMC article.
-
Fetuin-A mediates the difference in adipose tissue insulin resistance between young adult pakistani and norwegian patients with type 2 diabetes.BMC Endocr Disord. 2022 Aug 17;22(1):208. doi: 10.1186/s12902-022-01127-y. BMC Endocr Disord. 2022. PMID: 35978354 Free PMC article.
-
Diagnostic value of pentraxin 3 in respiratory tract infections: A meta-analysis.Medicine (Baltimore). 2020 Apr;99(14):e19532. doi: 10.1097/MD.0000000000019532. Medicine (Baltimore). 2020. PMID: 32243370 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

