Definition and treatment of arrhythmogenic cardiomyopathy: an updated expert panel report
- PMID: 31210398
- PMCID: PMC6685753
- DOI: 10.1002/ejhf.1534
Definition and treatment of arrhythmogenic cardiomyopathy: an updated expert panel report
Abstract
It is 35 years since the first description of arrhythmogenic right ventricular cardiomyopathy (ARVC) and more than 20 years since the first reports establishing desmosomal gene mutations as a major cause of the disease. Early advances in the understanding of the clinical, pathological and genetic architecture of ARVC resulted in consensus diagnostic criteria, which proved to be sensitive but not entirely specific for the disease. In more recent years, clinical and genetic data from families and the recognition of a much broader spectrum of structural disorders affecting both ventricles and associated with a propensity to ventricular arrhythmia have raised many questions about pathogenesis, disease terminology and clinical management. In this paper, we present the conclusions of an expert round table that aimed to summarise the current state of the art in arrhythmogenic cardiomyopathies and to define future research priorities.
Keywords: Arrhythmogenic cardiomyopathy; Arrhythmogenic right ventricular cardiomyopathy; Ventricular arrhythmias.
© 2019 The Authors. European Journal of Heart Failure © 2019 European Society of Cardiology.
Figures



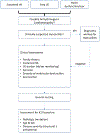
References
-
- Zorzi A, Perazzolo Marra M, Rigato I, De Lazzari M, Susana A, Niero A, Pilichou K, Migliore F, Rizzo S, Giorgi B, De Conti G, Sarto P, Serratosa L, Patrizi G, De Maria E, Pelliccia A, Basso C, Schiavon M, Bauce B, Iliceto S, Thiene G, Corrado D. Nonischemic left ventricular scar as a substrate of life-threatening ventricular arrhythmias and sudden cardiac death in competitive athletes. Circ Arrhythm Electrophysiol 2016;9:e004229. - PMC - PubMed
-
- Nucifora G, Muser D, Masci PG, Barison A, Rebellato L, Piccoli G, Daleffe E, Toniolo M, Zanuttini D, Facchin D, Lombardi M, Proclemer A. Prevalence and prognostic value of concealed structural abnormalities in patients with apparently idiopathic ventricular arrhythmias of left versus right ventricular origin: a magnetic resonance imaging study. Circ Arrhythm Electrophysiol 2014;7:456–462. - PubMed
-
- Pinto YM, Elliott PM, Arbustini E, Adler Y, Anastasakis A, Bohm M, Duboc D, Gimeno J, de Groote P, Imazio M, Heymans S, Klingel K, Komajda M, Limongelli G, Linhart A, Mogensen J, Moon J, Pieper PG, Seferovic PM, Schueler S, Zamorano JL, Caforio AL, Charron P. Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC working group on myocardial and pericardial diseases. Eur Heart J 2016;37:1850–1858. - PubMed
-
- McKoy G, Protonotarios N, Crosby A, Tsatsopoulou A, Anastasakis A, Coonar A, Norman M, Baboonian C, Jeffery S, McKenna WJ. Identification of a deletion in plakoglobin in arrhythmogenic right ventricular cardiomyopathy with palmoplantar keratoderma and woolly hair (Naxos disease). Lancet 2000;355:2119–2124. - PubMed
-
- Norgett EE, Hatsell SJ, Carvajal-Huerta L, Cabezas JC, Common J, Purkis PE, Whittock N, Leigh IM, Stevens HP, Kelsell DP. Recessive mutation in desmoplakin disrupts desmoplakin-intermediate filament interactions and causes dilated cardiomyopathy, woolly hair and keratoderma. Hum Mol Genet 2000;9:2761–2766. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

