Factors Affecting the Regeneration, via Organogenesis, and the Selection of Transgenic Calli in the Peach Rootstock Hansen 536 (Prunus persica × Prunus amygdalus) to Express an RNAi Construct against PPV Virus
- PMID: 31213013
- PMCID: PMC6631258
- DOI: 10.3390/plants8060178
Factors Affecting the Regeneration, via Organogenesis, and the Selection of Transgenic Calli in the Peach Rootstock Hansen 536 (Prunus persica × Prunus amygdalus) to Express an RNAi Construct against PPV Virus
Abstract
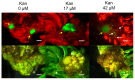
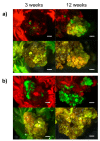
Prunus spp. is one of the most recalcitrant fruit tree species in terms of in vitro regeneration and transformation, mostly when mature tissues are used as explants. The present study describes the in vitro regeneration via indirect organogenesis, and Agrobacterium tumefaciens-mediated transformation of the peach rootstock Hansen 536 (Prunus persica × Prunus amygdalus) through the use of meristematic bulks (MBs) as starting explants. Efficient adventitious shoot regeneration was obtained when Hansen 536 MBs were cultured on an optimized medium consisting of modified McCown Woody Plant medium (WPM) enriched with 4.4 M 6-Benzyladenine (BA), 0.1 M 1-Naphthaleneacetic acid (NAA) and 6.0 g L-1 plant agar S1000 (B&V). MB slices were used later as starting explants for Agrobacterium-mediated transformation to introduce an RNAi construct "ihp35S-PPV194" against PPV virus. Transgenic events were identified by both green fluorescent protein (GFP) screening and kanamycin selection at different concentrations (0, 17 or 42 M). GFP-fluorescent proliferating callus lines were selected and confirmed to stably express the ihp35S-PPV194::eGFP gene construct by molecular analysis. Although shoot regeneration from these transgenic calli has not been obtained yet, this represents one of the few examples of successful attempts in peach genetic transformation from somatic tissues, and also serves as a useful in vitro system for future gene functional analysis in peach.
Keywords: GFP; PPV; Prunus spp.; RNAi; genetic transformation; organogenesis.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Adventitious Shoot Regeneration from In Vitro Leaf Explants of the Peach Rootstock Hansen 536.Plants (Basel). 2020 Jun 16;9(6):755. doi: 10.3390/plants9060755. Plants (Basel). 2020. PMID: 32560119 Free PMC article.
-
Peach (Prunus persica L.).Methods Mol Biol. 2015;1224:205-15. doi: 10.1007/978-1-4939-1658-0_17. Methods Mol Biol. 2015. PMID: 25416260
-
Comparison of regeneration capacity and Agrobacterium-mediated cell transformation efficiency of different cultivars and rootstocks of Vitis spp. via organogenesis.Sci Rep. 2019 Jan 24;9(1):582. doi: 10.1038/s41598-018-37335-7. Sci Rep. 2019. PMID: 30679725 Free PMC article.
-
Genetic Transformation in Peach (Prunus persica L.): Challenges and Ways Forward.Plants (Basel). 2020 Jul 31;9(8):971. doi: 10.3390/plants9080971. Plants (Basel). 2020. PMID: 32752031 Free PMC article. Review.
-
The use of transgenic fruit trees as a resistance strategy for virus epidemics: the plum pox (sharka) model.Virus Res. 2000 Nov;71(1-2):63-9. doi: 10.1016/s0168-1702(00)00188-x. Virus Res. 2000. PMID: 11137162 Review.
Cited by
-
Source of Explant and Light Spectrum Influence in Adventitious Shoot Regeneration of Prunus salicina Lindl. (Japanese plum).Plants (Basel). 2025 Jul 18;14(14):2230. doi: 10.3390/plants14142230. Plants (Basel). 2025. PMID: 40733467 Free PMC article.
-
Creating a novel petal regeneration system for function identification of colour gene of grape hyacinth.Plant Methods. 2021 Sep 16;17(1):94. doi: 10.1186/s13007-021-00794-7. Plant Methods. 2021. PMID: 34530873 Free PMC article.
-
Adventitious Shoot Regeneration from In Vitro Leaf Explants of the Peach Rootstock Hansen 536.Plants (Basel). 2020 Jun 16;9(6):755. doi: 10.3390/plants9060755. Plants (Basel). 2020. PMID: 32560119 Free PMC article.
-
Overview on Current Selectable Marker Systems and Novel Marker Free Approaches in Fruit Tree Genetic Engineering.Int J Mol Sci. 2024 Nov 6;25(22):11902. doi: 10.3390/ijms252211902. Int J Mol Sci. 2024. PMID: 39595971 Free PMC article. Review.
-
Efficient Plant Regeneration and Transient Genetic Transformation System of Prunus xueluoensis via an Agrobacterium-Mediated Method.Int J Mol Sci. 2025 Apr 10;26(8):3588. doi: 10.3390/ijms26083588. Int J Mol Sci. 2025. PMID: 40332136 Free PMC article.
References
-
- Petri C., Hily J.-M., Vann C., Dardick C., Scorza R. A high-throughput transformation system allows the regeneration of marker-free plum plants (Prunus domestica) Ann. Appl. Biol. 2011;159:302–315. doi: 10.1111/j.1744-7348.2011.00499.x. - DOI
-
- García-Almodóvar R.C., Clemente-Moreno M.J., Díaz-Vivancos P., Petri C., Rubio M., Padilla I.M.G., Ilardi V., Burgos L. Greenhouse evaluation confirms in vitro sharka resistance of genetically engineered h-UTR/P1 plum plants. Plant Cell Tissue Organ Cult. 2015;120:791–796. doi: 10.1007/s11240-014-0629-7. - DOI
-
- Hily J.-M., Ravelonandro M., Damsteegt V., Bassett C., Petri C., Liu Z., Scorza R. Plum Pox Virus Coat Protein Gene Intron-hairpin-RNA (ihpRNA) Constructs Provide Resistance to Plum Pox Virus in Nicotiana benthamiana and Prunus domestica. J. Am. Soc. Hortic. Sci. 2007;132:850–858. doi: 10.21273/JASHS.132.6.850. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

