Antibody-Based Delivery of Cytokine Payloads to Carbonic Anhydrase IX Leads to Cancer Cures in Immunocompetent Tumor-Bearing Mice
- PMID: 31213507
- PMCID: PMC6994256
- DOI: 10.1158/1535-7163.MCT-18-1301
Antibody-Based Delivery of Cytokine Payloads to Carbonic Anhydrase IX Leads to Cancer Cures in Immunocompetent Tumor-Bearing Mice
Abstract
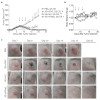
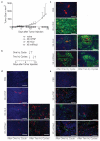
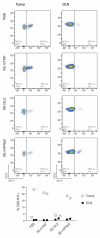
Antibody-cytokine fusion proteins can have the potential to increase the density and activity of subsets of leukocytes within the tumor mass. Here, we describe the design, production, and characterization of four novel antibody-cytokine fusion proteins directed against human carbonic anhydrase IX, a highly validated marker of hypoxia that is overexpressed in clear cell renal cell carcinoma and other malignancies. As immunomodulatory payloads we used TNF, IL2, IFNα2 (corresponding to products that are in clinical use), and IL12 (as this cytokine potently activates T cells and NK cells). Therapy experiments were performed in BALB/c mice, bearing CT26 tumors transfected with human carbonic anhydrase IX, in order to assess the performance of the fusion proteins in an immunocompetent setting. The biopharmaceuticals featuring TNF, IL2, or IL12 as payloads cured all mice in their therapy groups, whereas only a subset of mice was cured by the antibody-based delivery of IFNα2. Although the antibody fusion with TNF mediated a rapid hemorrhagic necrosis of the tumor mass, a slower regression of the neoplastic lesions (which continued after the last injection) was observed with the other fusion proteins, and treated mice acquired protective anticancer immunity. A high proportion of tumor-infiltrating CD8+ T cells was specific to the retroviral antigen AH1; however, the LGPGREYRAL peptide derived from human carbonic anhydrase IX was also present on tumor cells. The results described herein provide a rationale for the clinical use of fully human antibody-cytokine fusions specific to carbonic anhydrase IX.
©2019 American Association for Cancer Research.
Conflict of interest statement
Figures


Similar articles
-
Enhanced Therapeutic Activity of Non-Internalizing Small-Molecule-Drug Conjugates Targeting Carbonic Anhydrase IX in Combination with Targeted Interleukin-2.Clin Cancer Res. 2018 Aug 1;24(15):3656-3667. doi: 10.1158/1078-0432.CCR-17-3457. Epub 2018 Apr 24. Clin Cancer Res. 2018. PMID: 29691298 Free PMC article.
-
Carbonic Anhydrase IX in Renal Cell Carcinoma, Implications for Disease Management.Int J Mol Sci. 2020 Sep 28;21(19):7146. doi: 10.3390/ijms21197146. Int J Mol Sci. 2020. PMID: 32998233 Free PMC article. Review.
-
Chemically Defined Antibody- and Small Molecule-Drug Conjugates for in Vivo Tumor Targeting Applications: A Comparative Analysis.J Am Chem Soc. 2018 Feb 7;140(5):1617-1621. doi: 10.1021/jacs.7b13361. Epub 2018 Jan 27. J Am Chem Soc. 2018. PMID: 29342352 Free PMC article.
-
A 99mTc-Labeled Ligand of Carbonic Anhydrase IX Selectively Targets Renal Cell Carcinoma In Vivo.J Nucl Med. 2016 Jun;57(6):943-9. doi: 10.2967/jnumed.115.170514. Epub 2016 Feb 18. J Nucl Med. 2016. PMID: 26912427
-
G250: a carbonic anhydrase IX monoclonal antibody.Curr Oncol Rep. 2005 Mar;7(2):109-15. doi: 10.1007/s11912-005-0036-7. Curr Oncol Rep. 2005. PMID: 15717944 Review.
Cited by
-
Exosomal microRNA-210 is a potentially non-invasive biomarker for the diagnosis and prognosis of glioma.Oncol Lett. 2020 Mar;19(3):1967-1974. doi: 10.3892/ol.2020.11249. Epub 2020 Jan 7. Oncol Lett. 2020. PMID: 32194691 Free PMC article.
-
Utilizing Immunocytokines for Cancer Therapy.Antibodies (Basel). 2021 Mar 9;10(1):10. doi: 10.3390/antib10010010. Antibodies (Basel). 2021. PMID: 33803078 Free PMC article. Review.
-
A novel nanobody-based immunocytokine of a mutant interleukin-2 as a potential cancer therapeutic.AMB Express. 2024 Feb 9;14(1):19. doi: 10.1186/s13568-023-01648-2. AMB Express. 2024. PMID: 38337114 Free PMC article.
-
A Novel Fully-Human Potency-Matched Dual Cytokine-Antibody Fusion Protein Targets Carbonic Anhydrase IX in Renal Cell Carcinomas.Front Oncol. 2019 Nov 13;9:1228. doi: 10.3389/fonc.2019.01228. eCollection 2019. Front Oncol. 2019. PMID: 31799191 Free PMC article.
-
IL12 immune therapy clinical trial review: Novel strategies for avoiding CRS-associated cytokines.Front Immunol. 2022 Sep 20;13:952231. doi: 10.3389/fimmu.2022.952231. eCollection 2022. Front Immunol. 2022. PMID: 36203573 Free PMC article. Review.
References
-
- Murphy KWC. Janeway's Immunobiology. 9th ed. 2017.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

