Radiologic Modalities and Response Assessment Schemes for Clinical and Preclinical Oncology Imaging
- PMID: 31214510
- PMCID: PMC6558006
- DOI: 10.3389/fonc.2019.00471
Radiologic Modalities and Response Assessment Schemes for Clinical and Preclinical Oncology Imaging
Abstract
Clinical drug trials for oncology have resulted in universal protocols for medical imaging in order to standardize protocols for image procurement, radiologic interpretation, and therapeutic response assessment. In recent years, there has been increasing interest in using large animal models to study oncologic disease, though few standards currently exist for imaging of large animal models. This article briefly reviews medical imaging modalities, the current state-of-the-art in radiologic diagnostic criteria and response assessment schemes for evaluating therapeutic response and disease progression, and translation of radiologic imaging protocols and standards to large animal models of malignant disease.
Keywords: Radiology; imaging; large animal model; standards; trial.
Figures
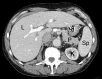
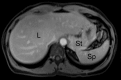
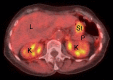
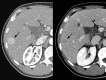
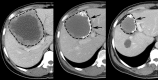
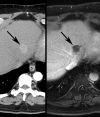
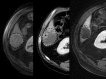
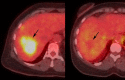
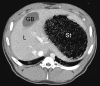
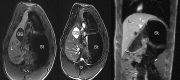
Similar articles
-
Trial design and reporting standards for intra-arterial cerebral thrombolysis for acute ischemic stroke.Stroke. 2003 Aug;34(8):e109-37. doi: 10.1161/01.STR.0000082721.62796.09. Epub 2003 Jul 17. Stroke. 2003. PMID: 12869717
-
The radiologic report: a realistic approach.Can Assoc Radiol J. 1995 Feb;46(1):19-22. Can Assoc Radiol J. 1995. PMID: 7834480 Review.
-
Radiological evaluation of the therapeutic response of malignant diseases: status quo, innovative developments and requirements for radiology.Rofo. 2014 Oct;186(10):927-36. doi: 10.1055/s-0034-1366741. Epub 2014 Aug 14. Rofo. 2014. PMID: 25122173 Review.
-
The current state of the art in three dimensional oncologic imaging: an overview.Int J Radiat Oncol Biol Phys. 1995 Dec 1;33(5):1029-39. doi: 10.1016/0360-3016(95)02003-9. Int J Radiat Oncol Biol Phys. 1995. PMID: 7493829 Review.
-
Imaging for oncologic staging and follow-up: review of current methods and novel approaches.Acta Biomed. 2008 Aug;79(2):85-91. Acta Biomed. 2008. PMID: 18788502 Review.
Cited by
-
Dual-energy CT for evaluating the tumor regression grade of gastric cancer after neoadjuvant chemotherapy.Eur Radiol. 2025 Sep;35(9):5748-5759. doi: 10.1007/s00330-025-11508-1. Epub 2025 Mar 18. Eur Radiol. 2025. PMID: 40100398
-
A Systematic Review and Meta-Analysis of 29 Studies Predicting Diagnostic Accuracy of CT, MRI, PET, and USG in Detecting Extracapsular Spread in Head and Neck Cancers.Cancers (Basel). 2024 Apr 10;16(8):1457. doi: 10.3390/cancers16081457. Cancers (Basel). 2024. PMID: 38672539 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources

