Heme oxygenase-2 protects against ischemic acute kidney injury: influence of age and sex
- PMID: 31215802
- PMCID: PMC6842883
- DOI: 10.1152/ajprenal.00085.2019
Heme oxygenase-2 protects against ischemic acute kidney injury: influence of age and sex
Abstract
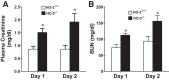
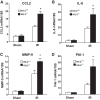
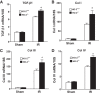
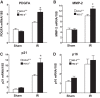
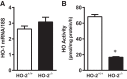
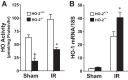
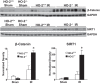
Heme oxygenase (HO) activity is exhibited by inducible (HO-1) and constitutive (HO-2) proteins. HO-1 protects against ischemic and nephrotoxic acute kidney injury (AKI). We have previously demonstrated that HO-2 protects against heme protein-induced AKI. The present study examined whether HO-2 is protective in ischemic AKI. Renal ischemia was imposed on young and aged HO-2+/+ and HO-2-/- mice. On days 1 and 2 after renal ischemia, there were no significant differences in renal function between young male HO-2+/+ and HO-2-/- mice, between young female HO-2+/+ and HO-2-/- mice, or between aged female HO-2+/+ and HO-2-/- mice. However, in aged male mice, HO-2 deficiency worsened renal function on days 1 and 2 after ischemic AKI, and, on day 2 after ischemia, such deficiency augmented upregulation of injury-related genes and worsened histological injury. Renal HO activity was markedly decreased in unstressed aged male HO-2-/- mice and remained so after ischemia, despite exaggerated HO-1 induction in HO-2-/- mice after ischemia. Such exacerbation of deficiency of HO-2 protein and HO activity may reflect phosphorylated STAT3, as activation of this proinflammatory transcription factor was accentuated early after ischemia in aged male HO-2-/- mice. This exacerbation may not reflect impaired induction of nephroprotectant genes, since the induction of HO-1, sirtuin 1, and β-catenin was accentuated in aged male HO-2-/- mice after ischemia. We conclude that aged male mice are hypersensitive to ischemic AKI and that HO-2 mitigates such sensitivity. We speculate that this protective effect of HO-2 may be mediated, at least in part, by suppression of phosphorylated STAT3-dependent signaling.
Keywords: acute kidney injury; age; biology of sex differences; heme oxygenase-2; phospho-STAT3.
Conflict of interest statement
J. D. Belcher and G. M. Vercellotti are consultants with and receive research funding from Mitobridge-Astellas. J. D. Belcher and G. M. Vercellotti receive research funding from CSL Behring.
Figures

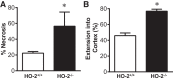

Similar articles
-
Age sensitizes the kidney to heme protein-induced acute kidney injury.Am J Physiol Renal Physiol. 2013 Feb 1;304(3):F317-25. doi: 10.1152/ajprenal.00606.2012. Epub 2012 Nov 28. Am J Physiol Renal Physiol. 2013. PMID: 23195679 Free PMC article.
-
6-Shogaol protects against ischemic acute kidney injury by modulating NF-κB and heme oxygenase-1 pathways.Am J Physiol Renal Physiol. 2019 Sep 1;317(3):F743-F756. doi: 10.1152/ajprenal.00182.2019. Epub 2019 Jul 17. Am J Physiol Renal Physiol. 2019. PMID: 31313953 Free PMC article.
-
Dual effect of hemin on renal ischemia-reperfusion injury.Biochem Biophys Res Commun. 2018 Sep 18;503(4):2820-2825. doi: 10.1016/j.bbrc.2018.08.046. Epub 2018 Aug 9. Biochem Biophys Res Commun. 2018. PMID: 30100067
-
Heme oxygenase-1 and acute kidney injury.Curr Opin Nephrol Hypertens. 2014 Jan;23(1):17-24. doi: 10.1097/01.mnh.0000437613.88158.d3. Curr Opin Nephrol Hypertens. 2014. PMID: 24275768 Free PMC article. Review.
-
Protective role of heme oxygenase-1 in renal ischemia.Antioxid Redox Signal. 2004 Oct;6(5):867-77. doi: 10.1089/ars.2004.6.867. Antioxid Redox Signal. 2004. PMID: 15345147 Review.
Cited by
-
Interaction among Hydrogen Sulfide and Other Gasotransmitters in Mammalian Physiology and Pathophysiology.Adv Exp Med Biol. 2021;1315:205-236. doi: 10.1007/978-981-16-0991-6_9. Adv Exp Med Biol. 2021. PMID: 34302694 Review.
-
The spike protein of SARS-CoV-2 induces heme oxygenase-1: Pathophysiologic implications.Biochim Biophys Acta Mol Basis Dis. 2022 Mar 1;1868(3):166322. doi: 10.1016/j.bbadis.2021.166322. Epub 2021 Dec 14. Biochim Biophys Acta Mol Basis Dis. 2022. PMID: 34920080 Free PMC article.
-
Rodent models of AKI and AKI-CKD transition: an update in 2024.Am J Physiol Renal Physiol. 2024 Apr 1;326(4):F563-F583. doi: 10.1152/ajprenal.00402.2023. Epub 2024 Feb 1. Am J Physiol Renal Physiol. 2024. PMID: 38299215 Free PMC article. Review.
-
Heme Oxygenase 1: A Defensive Mediator in Kidney Diseases.Int J Mol Sci. 2021 Feb 18;22(4):2009. doi: 10.3390/ijms22042009. Int J Mol Sci. 2021. PMID: 33670516 Free PMC article. Review.
-
Is Environmental Cadmium Exposure Causally Related to Diabetes and Obesity?Cells. 2023 Dec 30;13(1):83. doi: 10.3390/cells13010083. Cells. 2023. PMID: 38201287 Free PMC article. Review.
References
-
- Adachi T, Ishikawa K, Hida W, Matsumoto H, Masuda T, Date F, Ogawa K, Takeda K, Furuyama K, Zhang Y, Kitamuro T, Ogawa H, Maruyama Y, Shibahara S. Hypoxemia and blunted hypoxic ventilatory responses in mice lacking heme oxygenase-2. Biochem Biophys Res Commun 320: 514–522, 2004. doi:10.1016/j.bbrc.2004.05.195. - DOI - PubMed
-
- Aghakhani Chegeni S, Rahimzadeh M, Montazerghaem H, Khayatian M, Dasturian F, Naderi N. Preliminary report on the association between STAT3 polymorphisms and susceptibility to acute kidney injury after cardiopulmonary bypass. Biochem Genet 56: 627–638, 2018. doi:10.1007/s10528-018-9865-6. - DOI - PubMed
-
- Agúndez JA, García-Martín E, Martínez C, Benito-León J, Millán-Pascual J, Díaz-Sánchez M, Calleja P, Pisa D, Turpín-Fenoll L, Alonso-Navarro H, Pastor P, Ortega-Cubero S, Ayuso-Peralta L, Torrecillas D, García-Albea E, Plaza-Nieto JF, Jiménez-Jiménez FJ. Heme oxygenase-1 and 2 common genetic variants and risk for multiple sclerosis. Sci Rep 6: 20830, 2016. doi:10.1038/srep20830. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

