Combinatorial diversity of Syk recruitment driven by its multivalent engagement with FcεRIγ
- PMID: 31216232
- PMCID: PMC6743456
- DOI: 10.1091/mbc.E18-11-0722
Combinatorial diversity of Syk recruitment driven by its multivalent engagement with FcεRIγ
Abstract
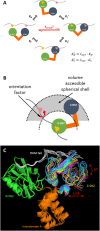
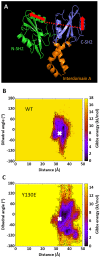
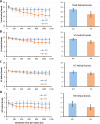
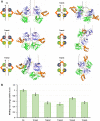
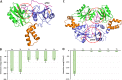
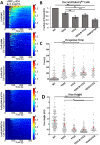
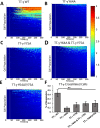
Syk/Zap70 family kinases are essential for signaling via multichain immune-recognition receptors such as tetrameric (αβγ2) FcεRI. Syk activation is generally attributed to cis binding of its tandem SH2 domains to dual phosphotyrosines within FcεRIγ-ITAMs (immunoreceptor tyrosine-based activation motifs). However, the mechanistic details of Syk docking on γ homodimers are unresolved. Here, we estimate that multivalent interactions for WT Syk improve cis-oriented binding by three orders of magnitude. We applied molecular dynamics (MD), hybrid MD/worm-like chain polymer modeling, and live cell imaging to evaluate relative binding and signaling output for all possible cis and trans Syk-FcεRIγ configurations. Syk binding is likely modulated during signaling by autophosphorylation on Y130 in interdomain A, since a Y130E phosphomimetic form of Syk is predicted to lead to reduced helicity of interdomain A and alter Syk's bias for cis binding. Experiments in reconstituted γ-KO cells, whose γ subunits are linked by disulfide bonds, as well as in cells expressing monomeric ITAM or hemITAM γ-chimeras, support model predictions that short distances between γ ITAM pairs are required for trans docking. We propose that the full range of docking configurations improves signaling efficiency by expanding the combinatorial possibilities for Syk recruitment, particularly under conditions of incomplete ITAM phosphorylation.
Figures

References
-
- Abraham MJ, Murtola T, Schulz R, Páll S, Smith JC, Hess B, Lindahl E. (2015). GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX , 19–25.
-
- Aqvist J, Medina C, Samuelsson JE. (1994). A new method for predicting binding affinity in computer-aided drug design. Protein Eng , 385–391. - PubMed
-
- Åqvist J, Wennerström P, Nervall M, Bjelic S, Brandsdal BO. (2004). Molecular dynamics simulations of water and biomolecules with a Monte Carlo constant pressure algorithm. Chem Phys Lett , 288–294.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

